Phenol Chemical Structure Functional Group
SKU: 68845NV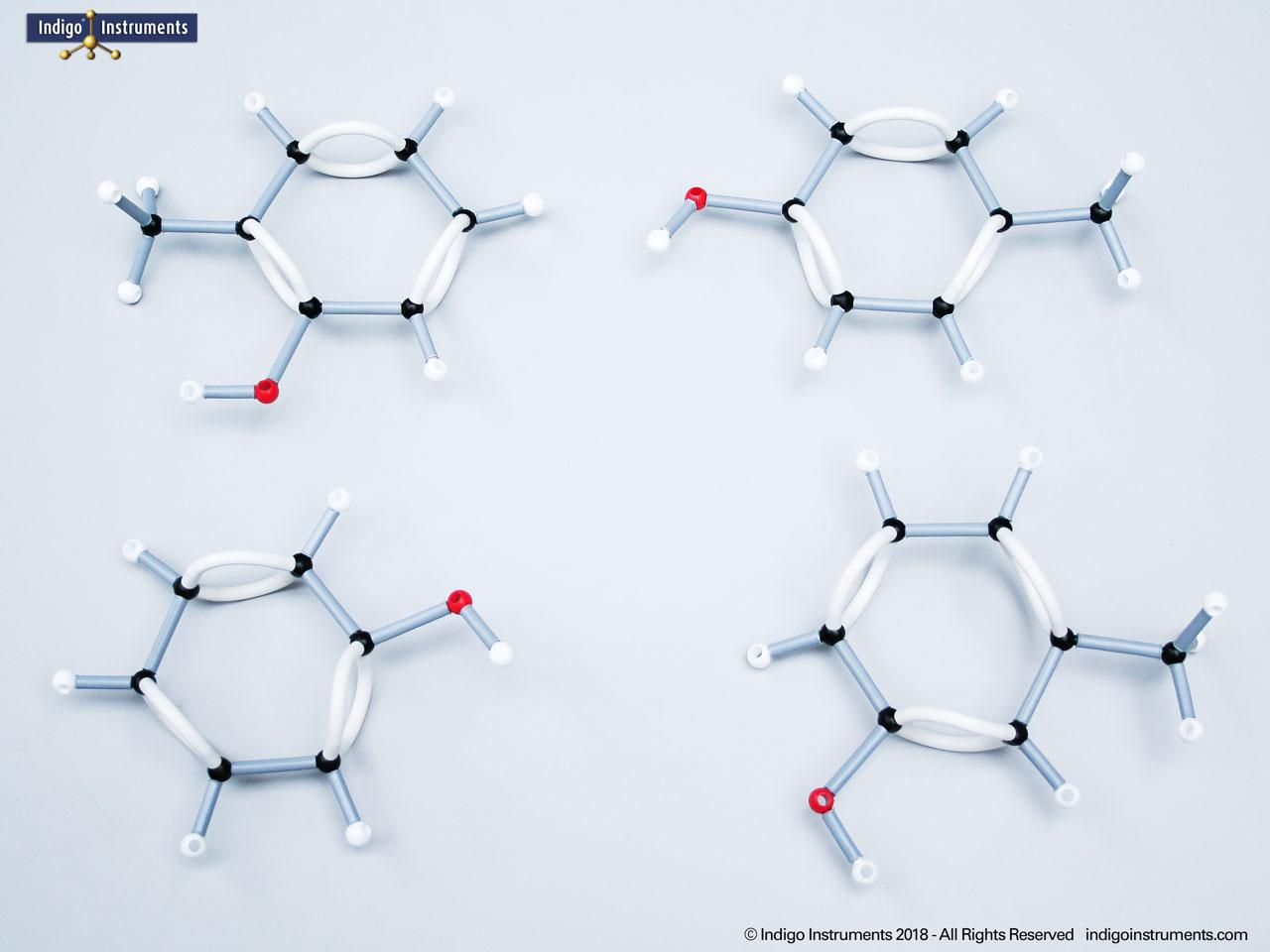
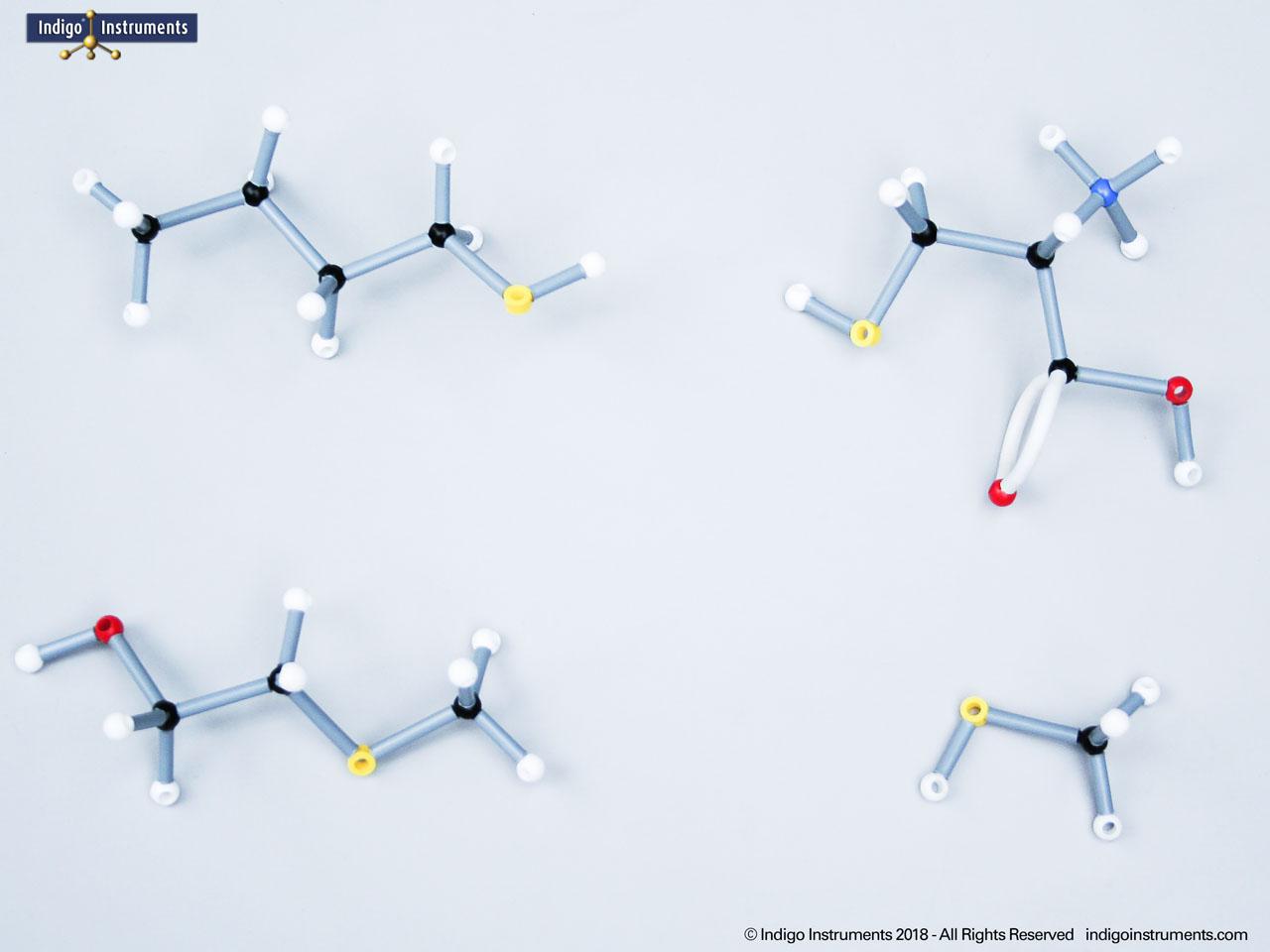
Examine phenols with Orbit molecular models. Learn phenol structure, functional groups, acidity, and biological significance for chemistry, biology, and medical students.
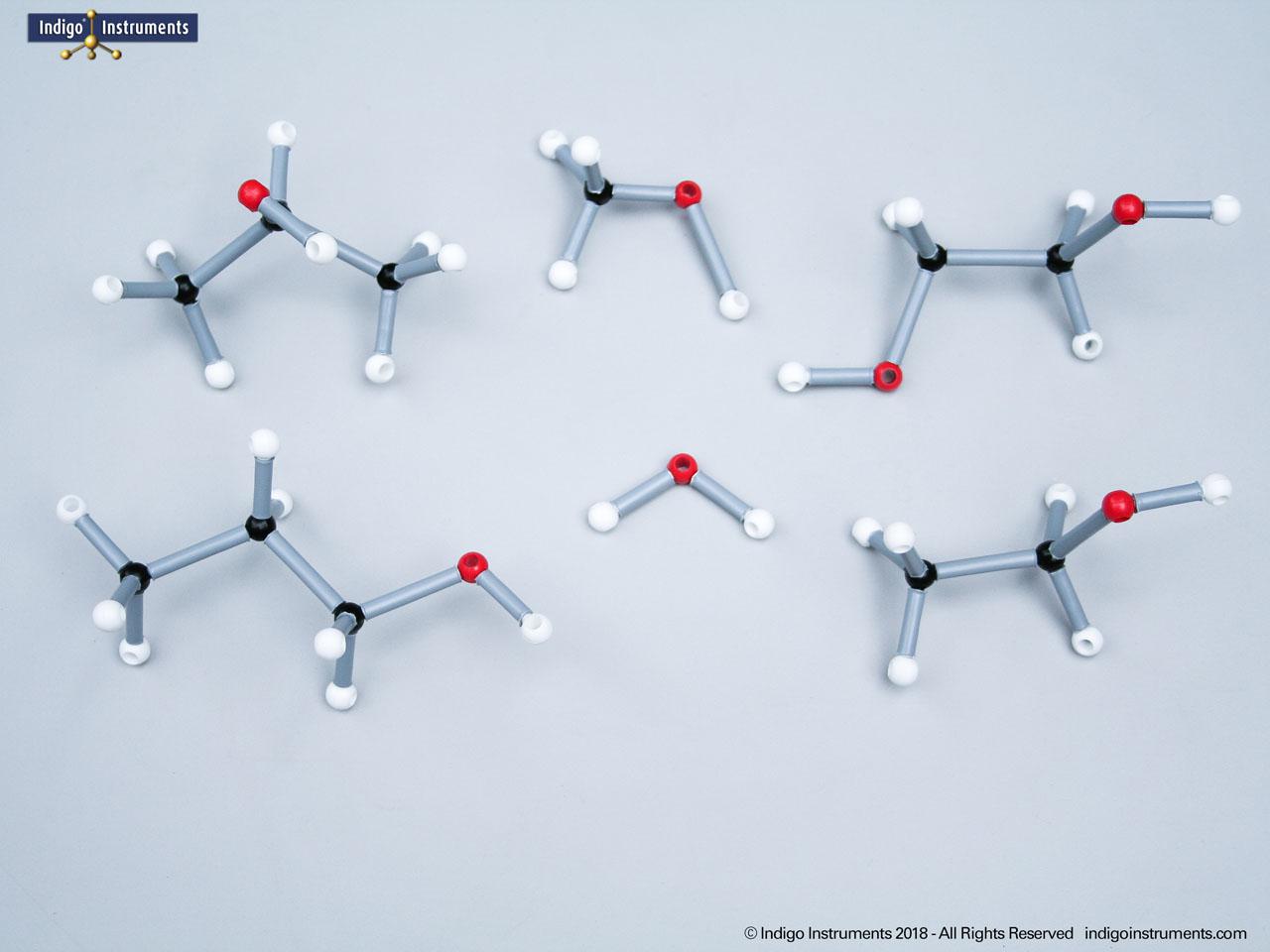
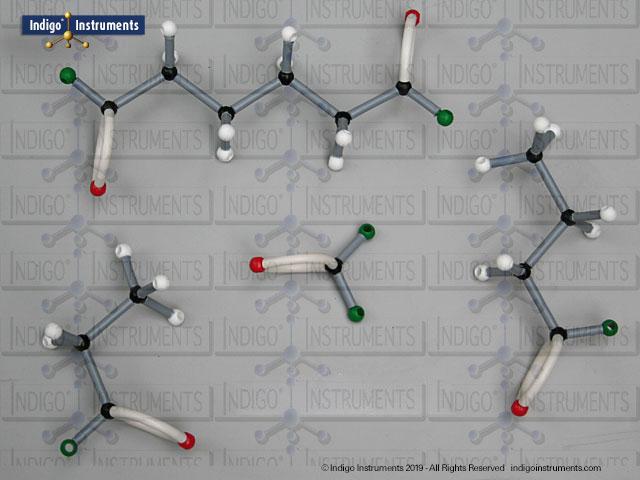
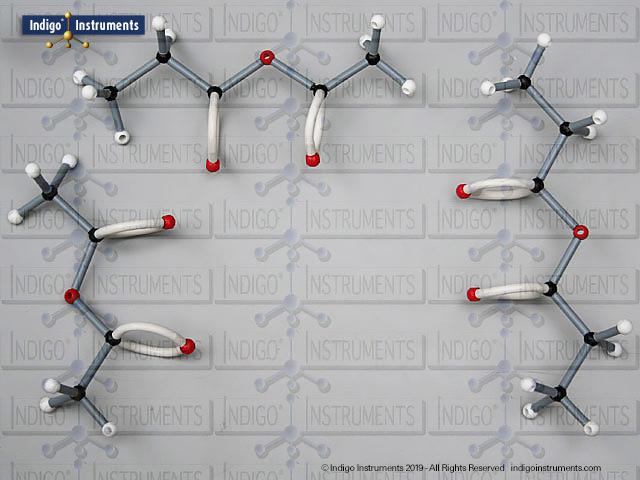
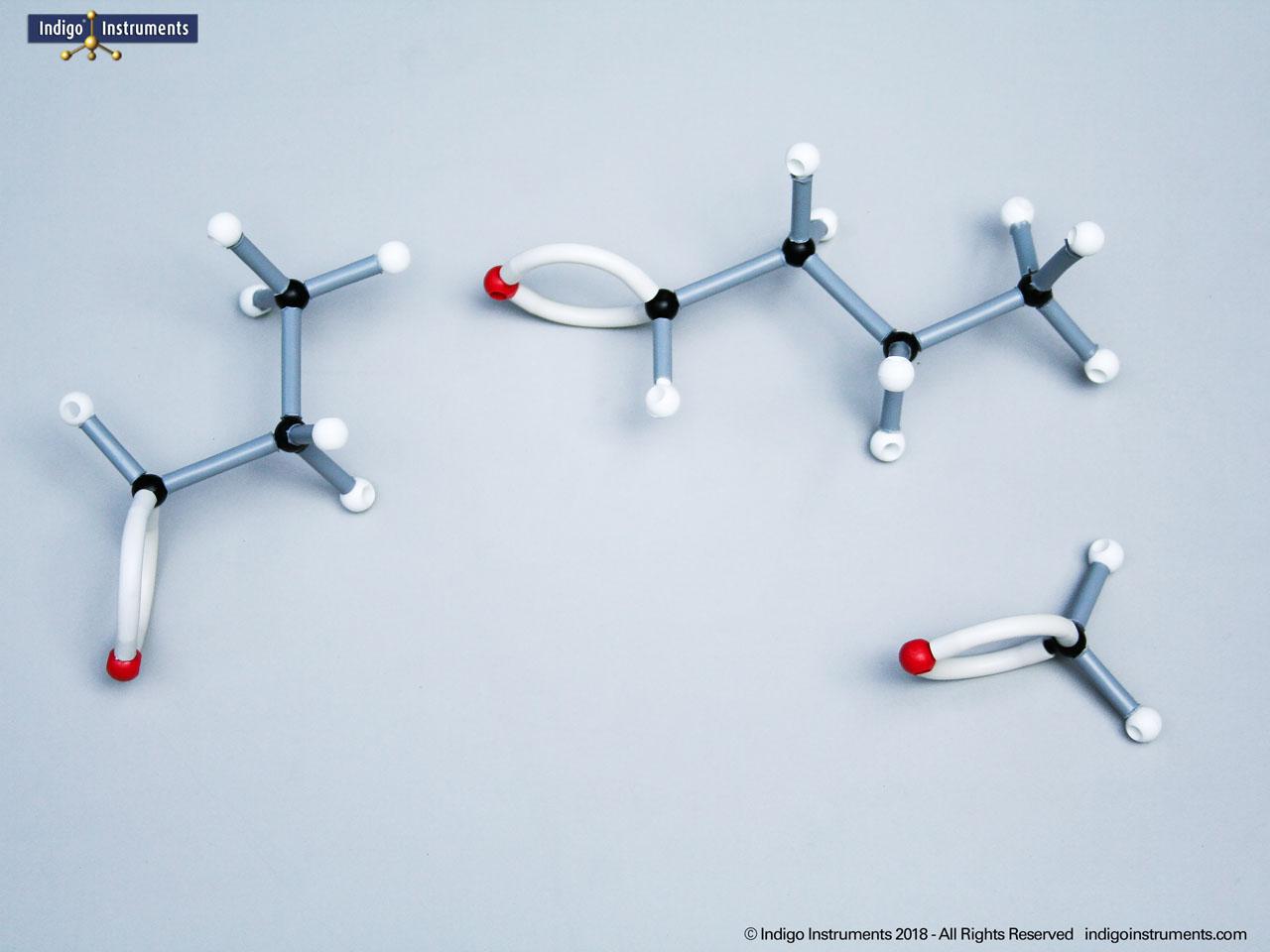
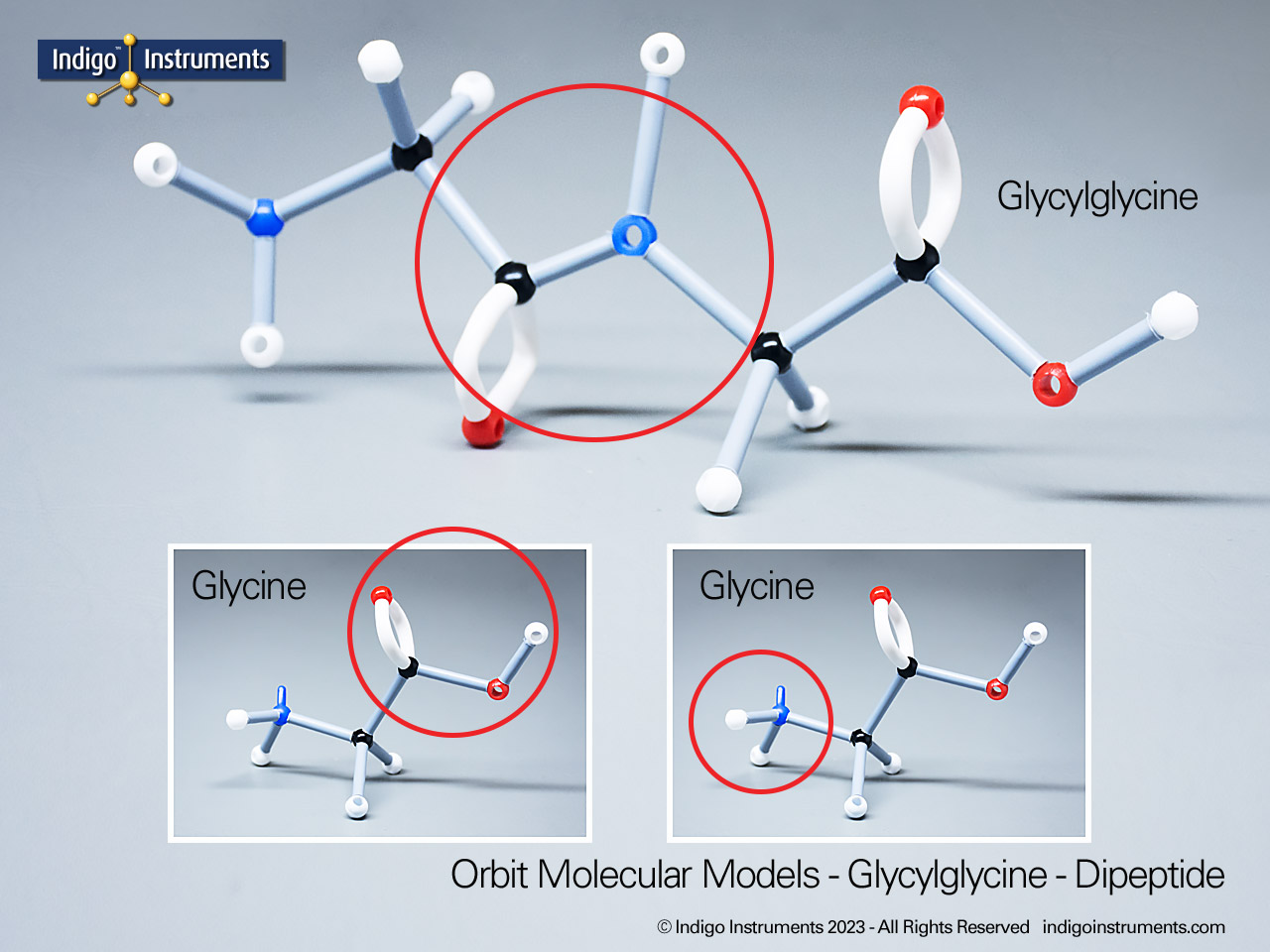
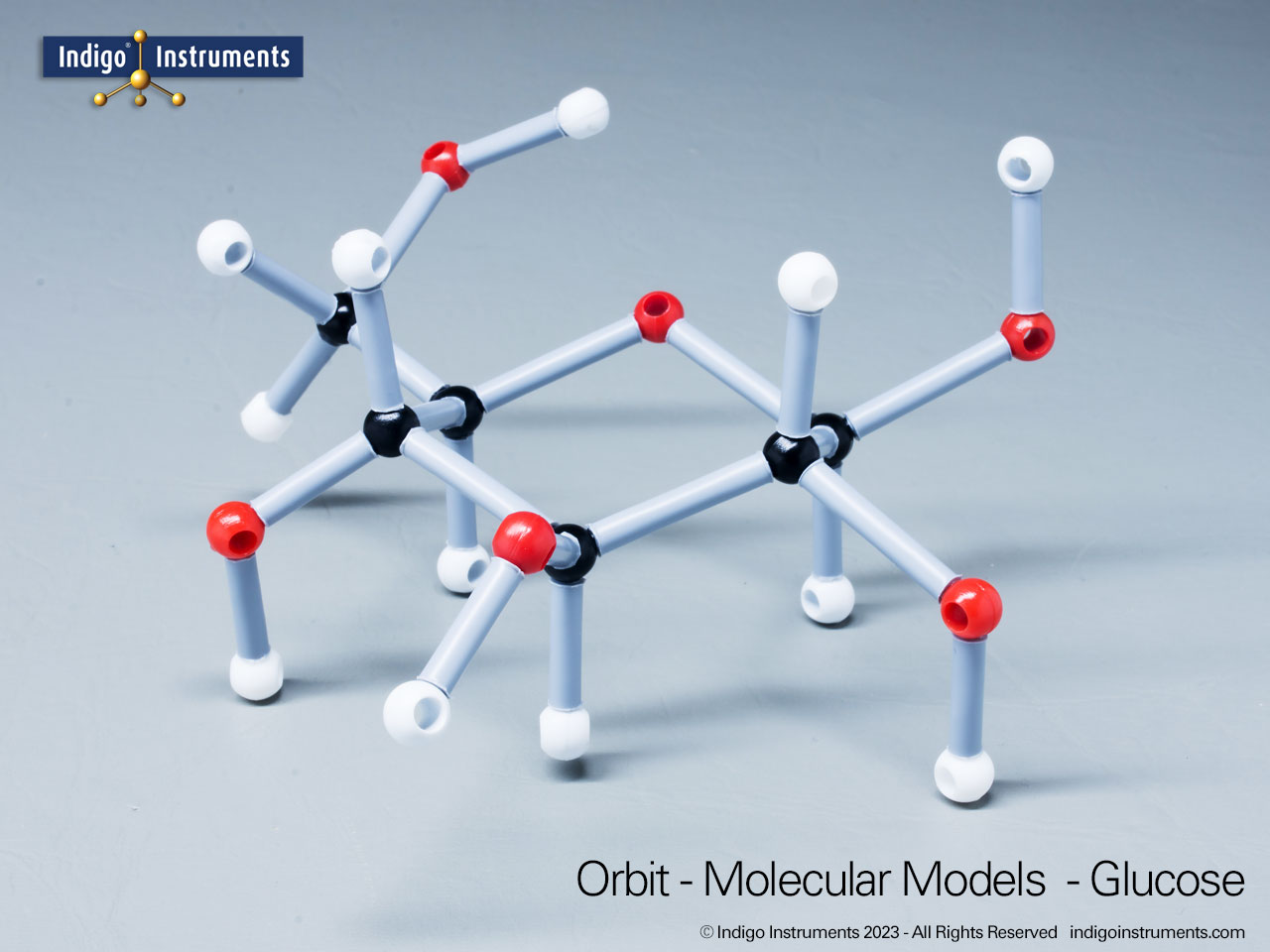
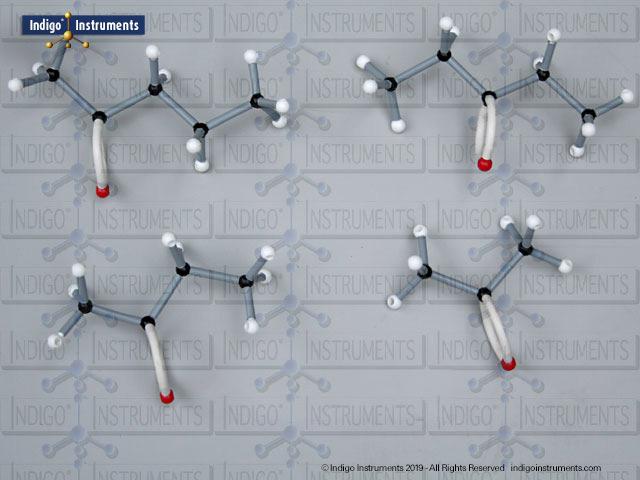
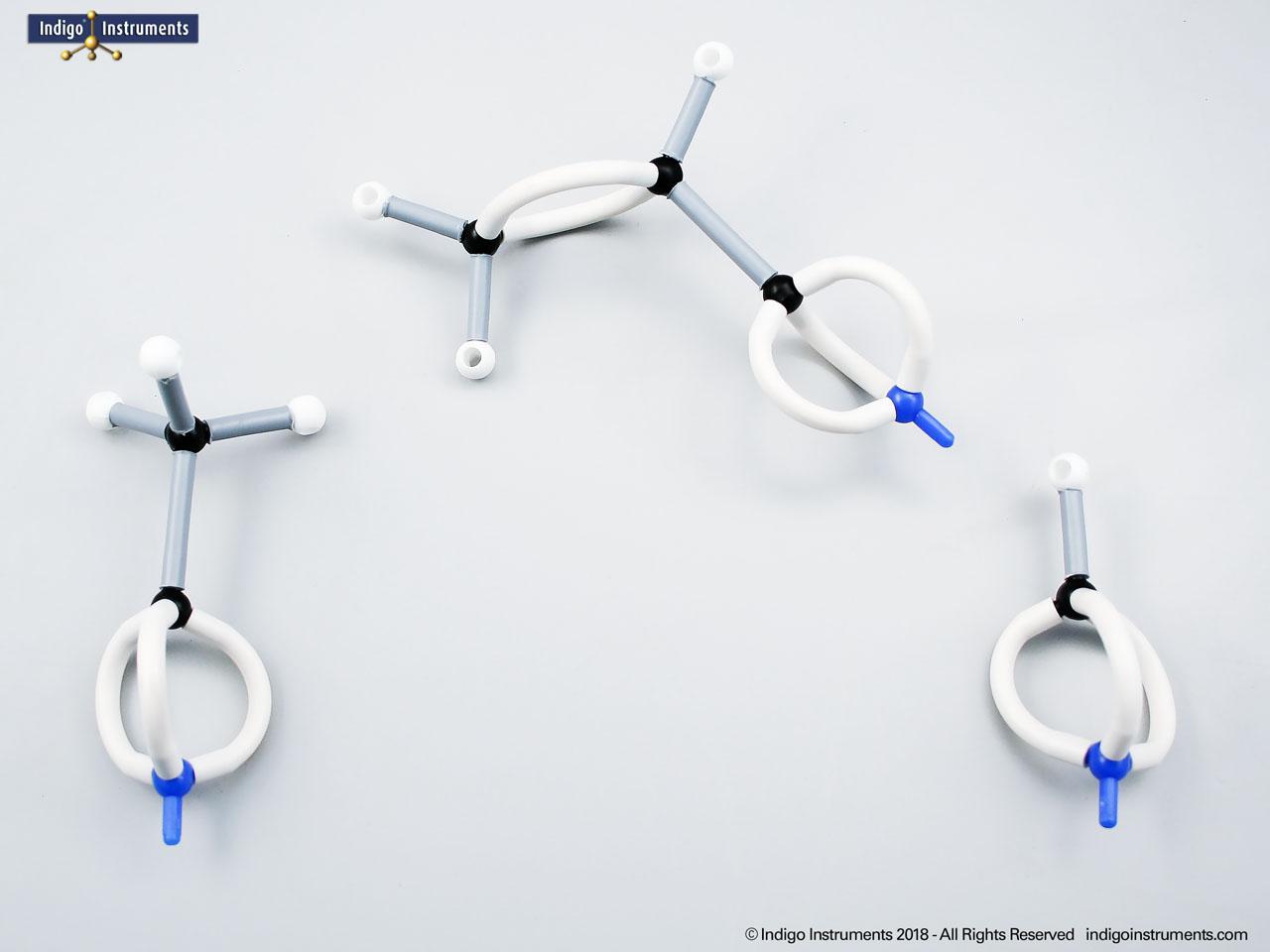
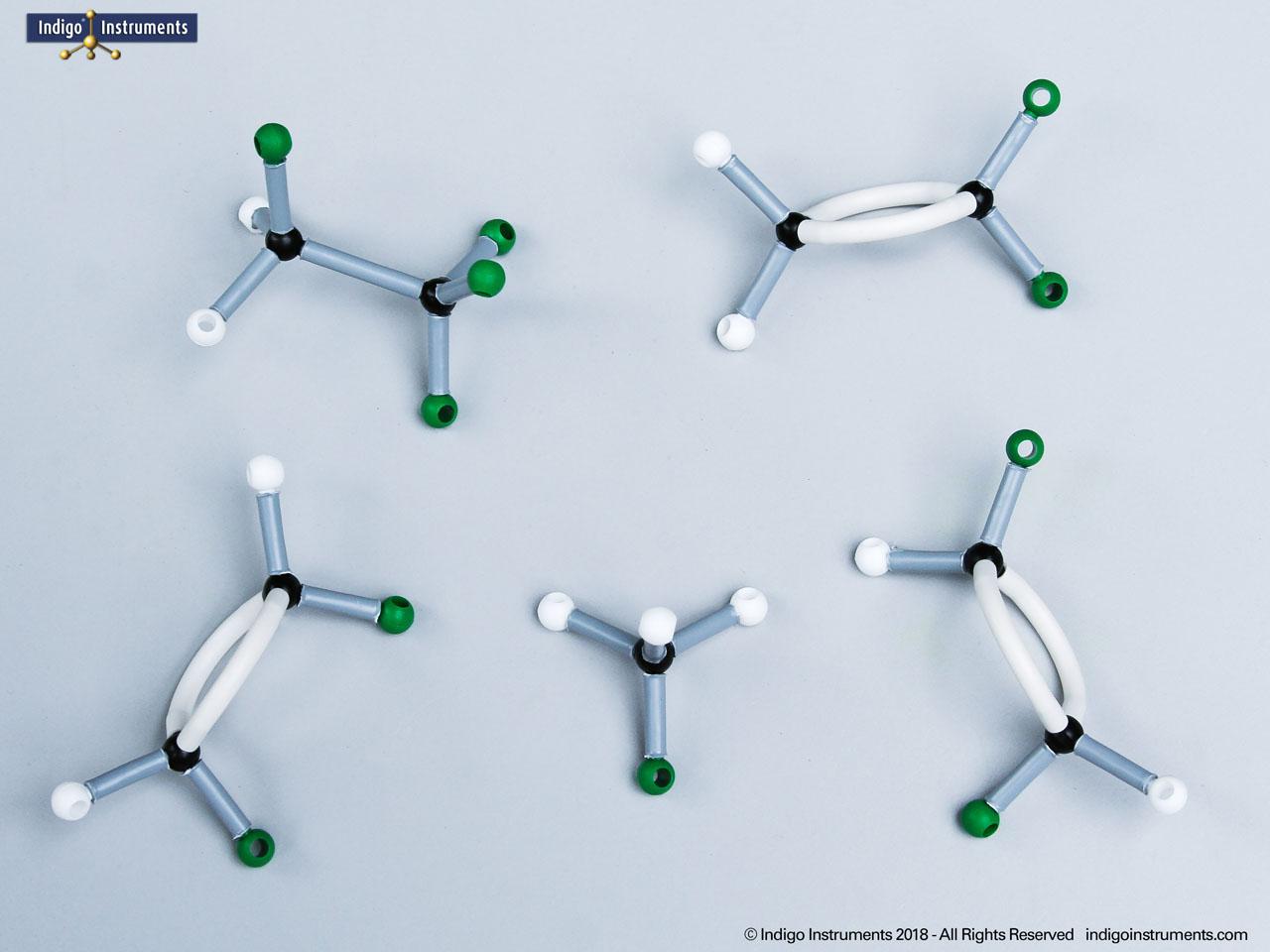
Phenols are aromatic compounds in which a hydroxyl group (-OH) is directly bonded to a benzene ring. This unique arrangement gives phenols chemical and biological properties distinct from both alcohols and simple aromatics. In organic chemistry, phenols are studied for their reactivity, acidity, and role in electrophilic aromatic substitution. In biology and medicine, phenols are essential in natural products, antiseptics, and pharmaceuticals, with applications ranging from antioxidants to anesthetics. Using a phenol molecular model allows students to visualize the aromatic π-system, hydrogen bonding potential, and the influence of resonance on acidity, critical for mastering phenol chemistry in both academic and applied settings.
Indigo Instruments has stocked genuine Cochranes of Oxford molecular model parts for 30+ years. Customize or replenish any model set/kit we have ever sold!