Hydrogen Peroxide Test Strips
Buy Indigo® hydrogen peroxide test strips for your disinfectant solution monitoring for food & beverages; pharmaceuticals; medical sterilization; aseptic packaging; industrial cleaning; & public health. We provide Certificates of Analysis & Stability & SDS Statements on-line; Certificates of Conformance on request.
Choose from 4 Indigo® H2O2 test strips for your specific disinfection & sanitizing needs. All Indigo® disinfectant/sanitizer test strips come with 100 test strips/vial and a minimum guaranteed 3 year shelf life (from date of manufacture). Our delivered costs beat "free" shipping"!
Optional next day delivery in any quantity of most items. Call us toll free at (877)746-4764 for immediate assistance (8-4 EST M-F). Emailed questions are typically answered within an hour.
Hydrogen Peroxide Disinfectant
Hydrogen peroxide is produced by our own immune system's white blood cells to eliminate invading micro-organisms. Excess peroxide is neutralized by enzymes such as catalase or super oxide dismutase (SOD) our own natural internal antioxidizers. It is used as a chemical disinfectant for its microbiocidal activity against bacteria, yeasts, fungi, viruses, and spores. Of the commonly used disinfectants, hydrogen peroxide may be more effective against Staphyloccocus aureus and Pseudomonas aeruginosa biofilms than quat chemical disinfectants.
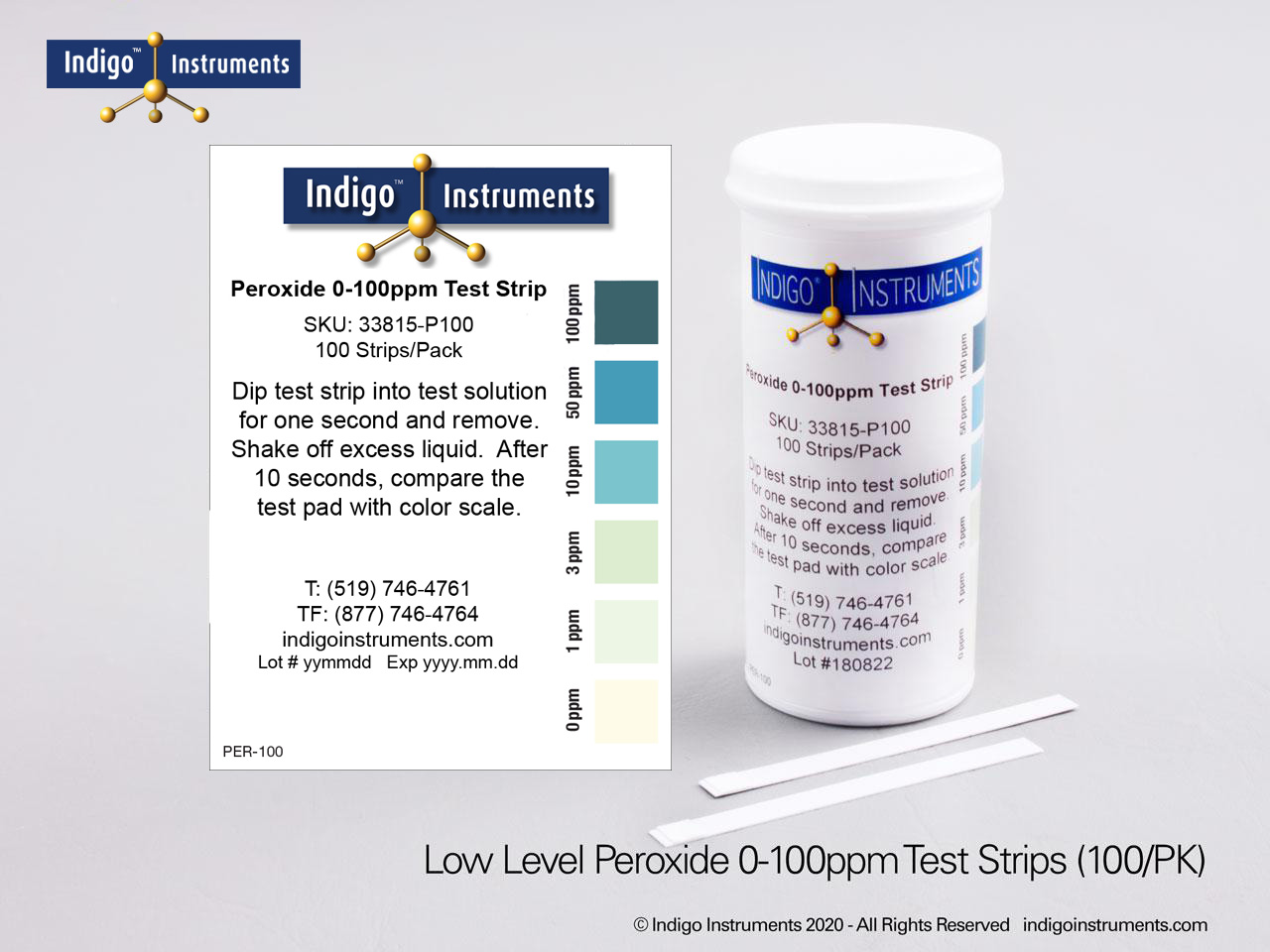
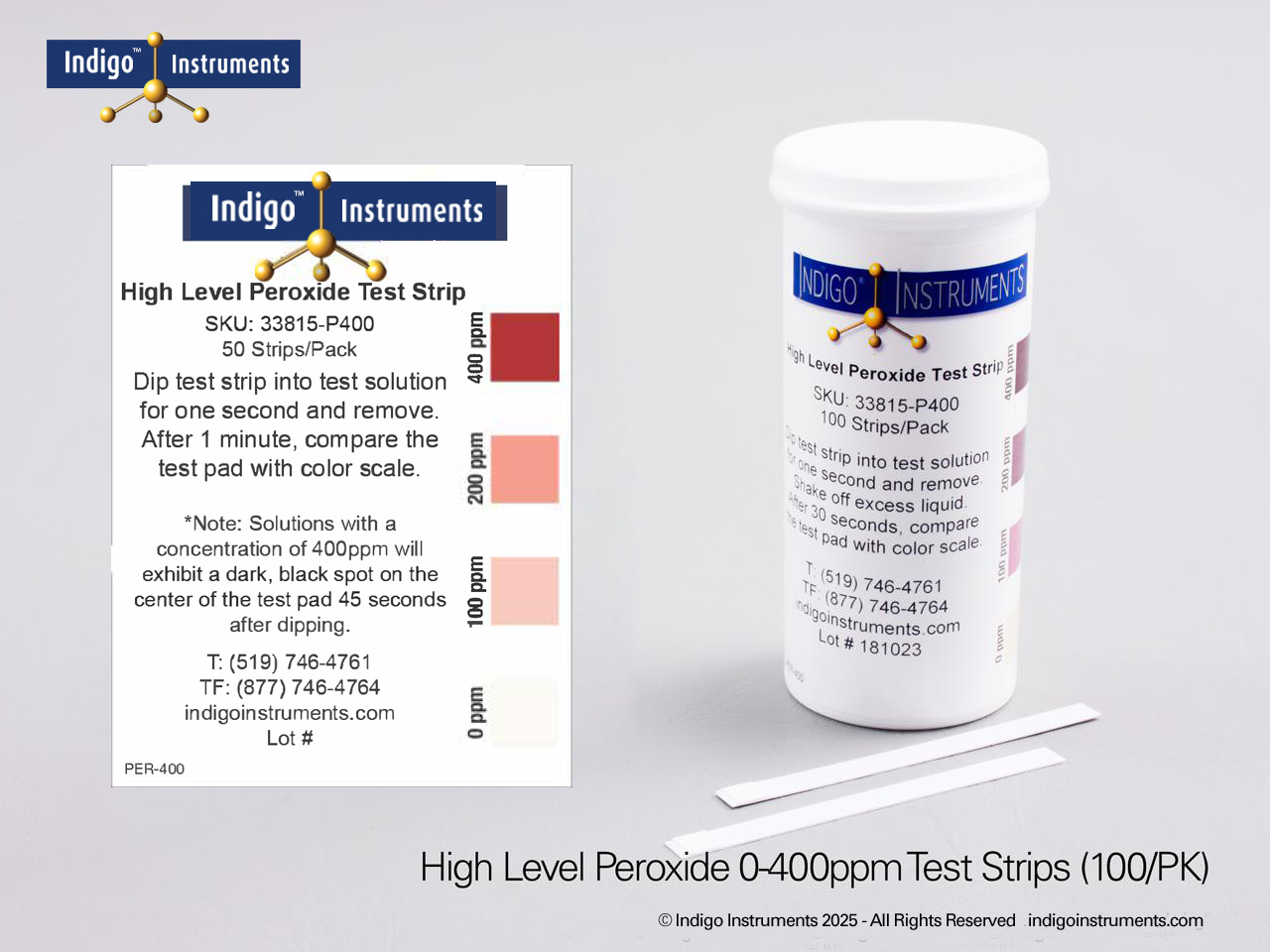
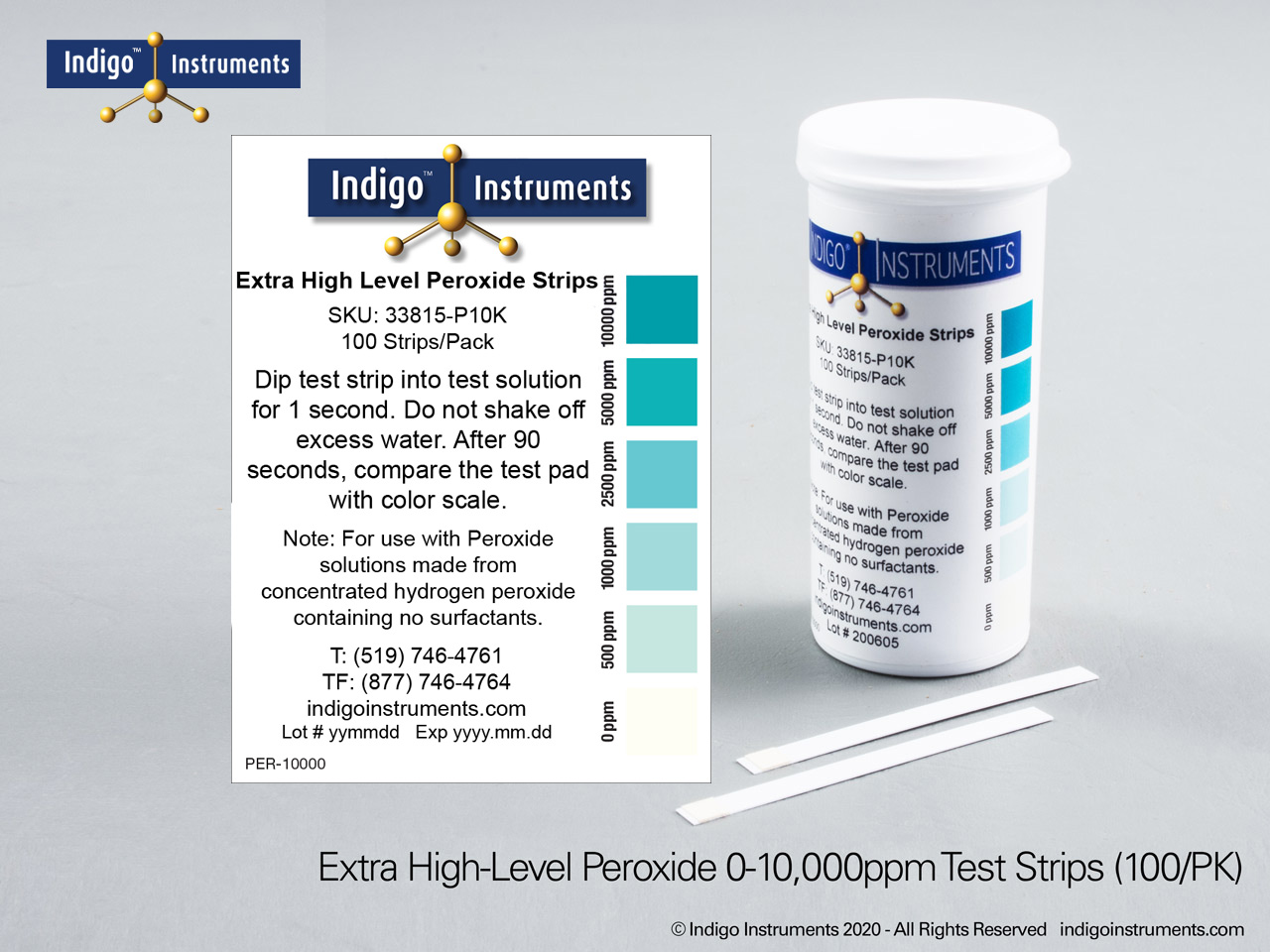
Indigo® Hydrogen Peroxide Test Strips
- 0-100ppm: 0-1-3-10-50-100ppm; read results in 10 seconds
- 0-400ppm: 0-100-200-300-400ppm; read results in 30 seconds
- 0-10000ppm: 0-500-1000-2500-5000-10000ppm; read results in 90 seconds
- 2-10%: timed results ~ w/ concentration; confirm 30-35% food grade dilutions
Common Features of Indigo® H2O2 Test Strips
- clear instructions with labelled color chart
- indicators on high contrast plastic for accurate readings
- stiff plastic strips ideal for dipping into tubes, etc.
- 3 year guaranteed shelf life
- Certificate of Analysis & Stability Statement, SDS online;
Certificate of Conformance on request.
Note: All test strips Made in the USA & are Q/A'd against NIST standards.