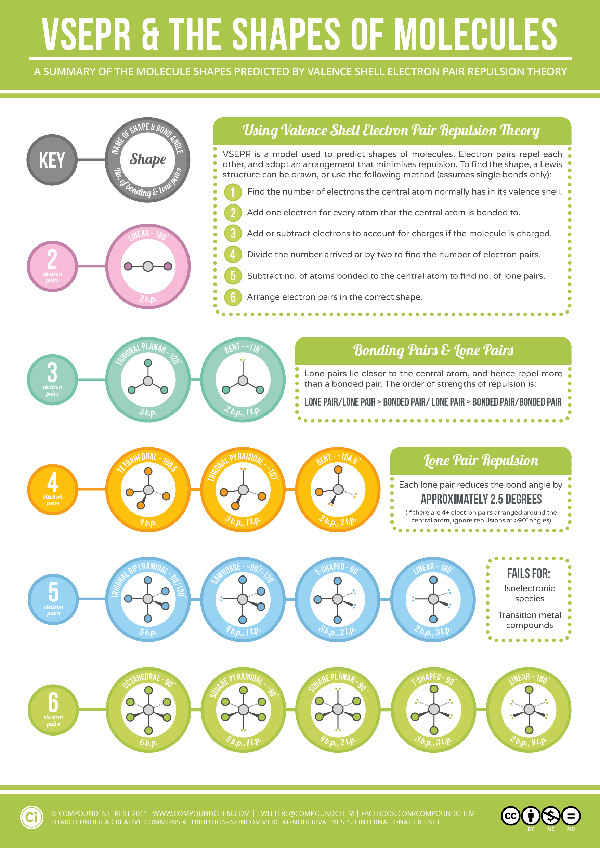
Molecular Geometry-VSEPR
VSEPR theory molecular models for teaching electron pair repulsion & shape of molecules.
Find the Indigo® VSEPR model kit that fits your classroom or study needs. Whether you want the flexibility of the Unit style to explore any geometry, or the student-friendly Orbit style for hands-on learning, there’s a model kit to bring molecular shapes to life. Start building your understanding of molecular geometry today!
Orbit Style VSEPR Theory Molecular Models
68823W (advanced kit): this is the most diverse kit. It comes with 15 10mm diameter black central atoms, a mixture of red and white univalent atoms (two-color scheme for visual interest) & black & white paddles to represent electron pairs. Several students can build the 15 most common standard VSEPR geometries for side by side comparison.
68820W (basic student kit): this kit has 5 10mm central atoms to illustrate the basic shapes of 5 typical compounds. Affordable and compact, this kit is designed primarily for individual student use.
68845NV basic organic chemistry student set: this is our most economical student set for organic chemistry. While intended for college/university level, it can in fact do all the VSEPR shapes shown in the 68823W VSEPR kit, just not all at once.
Note: Each of the above kits can be augmented with additional atoms, orbitals or bonds as listed in the Orbit Components page.
Unit Style VSEPR Theory Class Demonstration Molecular Models
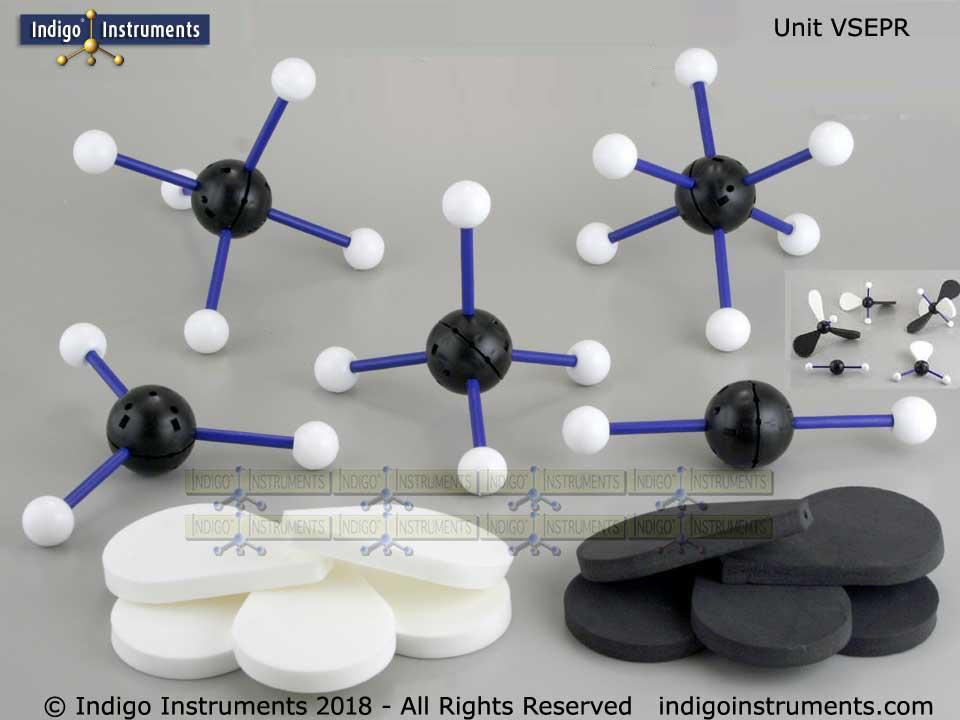
69182 (5 variable models kit): this versatile kit builds 5 models at a time. It comes with 5 diameter black atoms, 20 smaller white atoms, and 6 each of black and white “paddles” representing lone electron pairs. The black central atoms are configurable to any geometry for VSEPR teaching. The small white atoms can be used all at once or replaced by paddles to explore lone pair effects. Central atoms are 45?mm in diameter, ideal for teacher's use in classroom demonstrations.
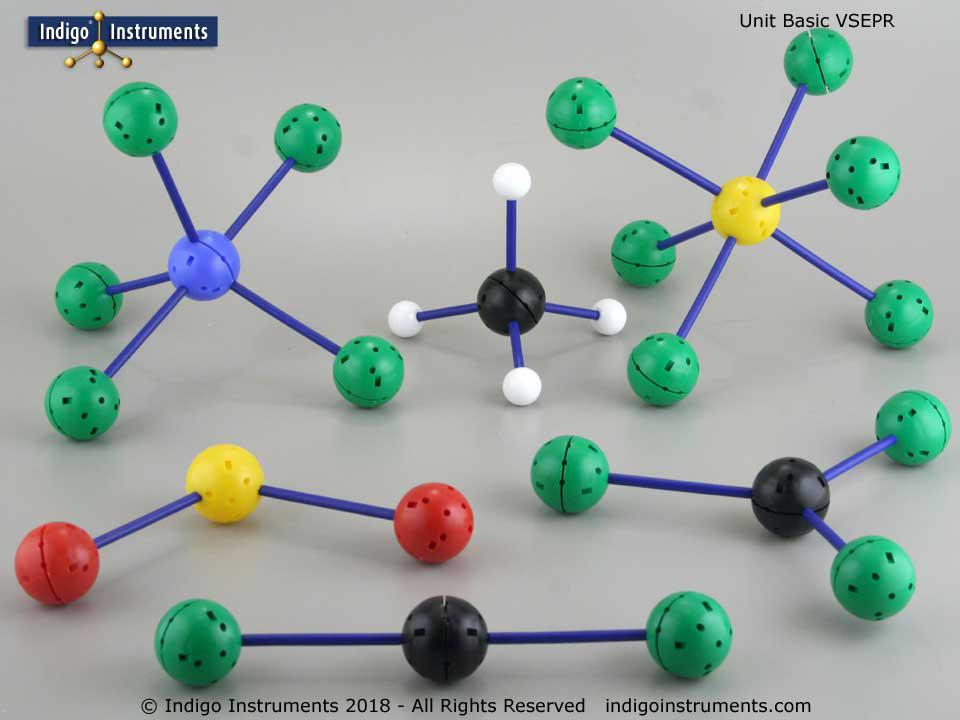
69180 (basic classroom version): this kit features 3 black, 2 yellow, and 1 blue central atoms combined with a variety of other atoms (white, green, red) to represent specific compounds. It does not include white or black paddles. This kit is excellent for teaching representative molecules without the full flexibility of 69182.
Note: Each of the above kits can be augmented with additional atoms, orbitals or bonds as listed in the Unit Components page.
Molymod Style VSEPR Theory Molecular Models
We do not stock a dedicated teacher's VSEPR theory model kit. However, you can use any of our Molymod student sets & augment them with parts from the Molymod Components page.