Quaternary Ammonium Disinfectant Compounds
Buy Indigo® quaternary ammonium compound test strips for for your disinfectant solution monitoring in restaurants, hospitals, organic farming & more. We provide Certificates of Analysis & Stability & SDS Statements on-line; Certificates of Conformance on request.
Choose from 4 Indigo® Quat test strips for your specific disinfection & sanitizing needs. All Indigo® disinfectant/sanitizer test strips come with 100 test strips/vial and a minimum guaranteed 3 year shelf life (from date of manufacture). Our delivered costs beat "free" shipping"!
Optional next day delivery in any quantity of most items. Call us toll free at (877)746-4764 for immediate assistance (8-4 EST M-F). Emailed questions are typically answered within an hour.
Quaternary Ammonium Compounds
Quats are effective germ-killers and a key ingredient in many cleaning, disinfection & infection control products. They are found in every day products such as household toilet cleaners, antibacterial hand soaps, dish washing liquids, baby wipes, body washes, disinfectant sprays & more. Quats are especially effective on hard, non-porous environmental surfaces. They are used in hospitals, long-term care facilities, households & food processing plants to name a few.
Quaternary ammonium compounds (quaternary ammonium salts) are cationic biocides (positively charged surface-active agents) that are more chemically complex than other disinfectants. Their name derives from the central nitrogen (quaternary ammonia) that is bound to four organic groups. They interfere with the metabolism of Gram-negative & Gram-positive vegetative bacteria and lipophilic viruses.
They have several advantages in the fight against antimicrobial resistance. Unlike antibiotics, disinfectants like quats are non-specific in their targeting of microorganisms. Their amphiphilic nature (having both hydrophobic & hydrophilic properties) allows for enhanced binding affinity that can disrupt bacterial cell membranes causing cell lysis.
Whatever formulation of quaternary ammonium you use, Indigo® quat test strips can confirm your disinfectant solutions are correctly diluted for optimum effectiveness.
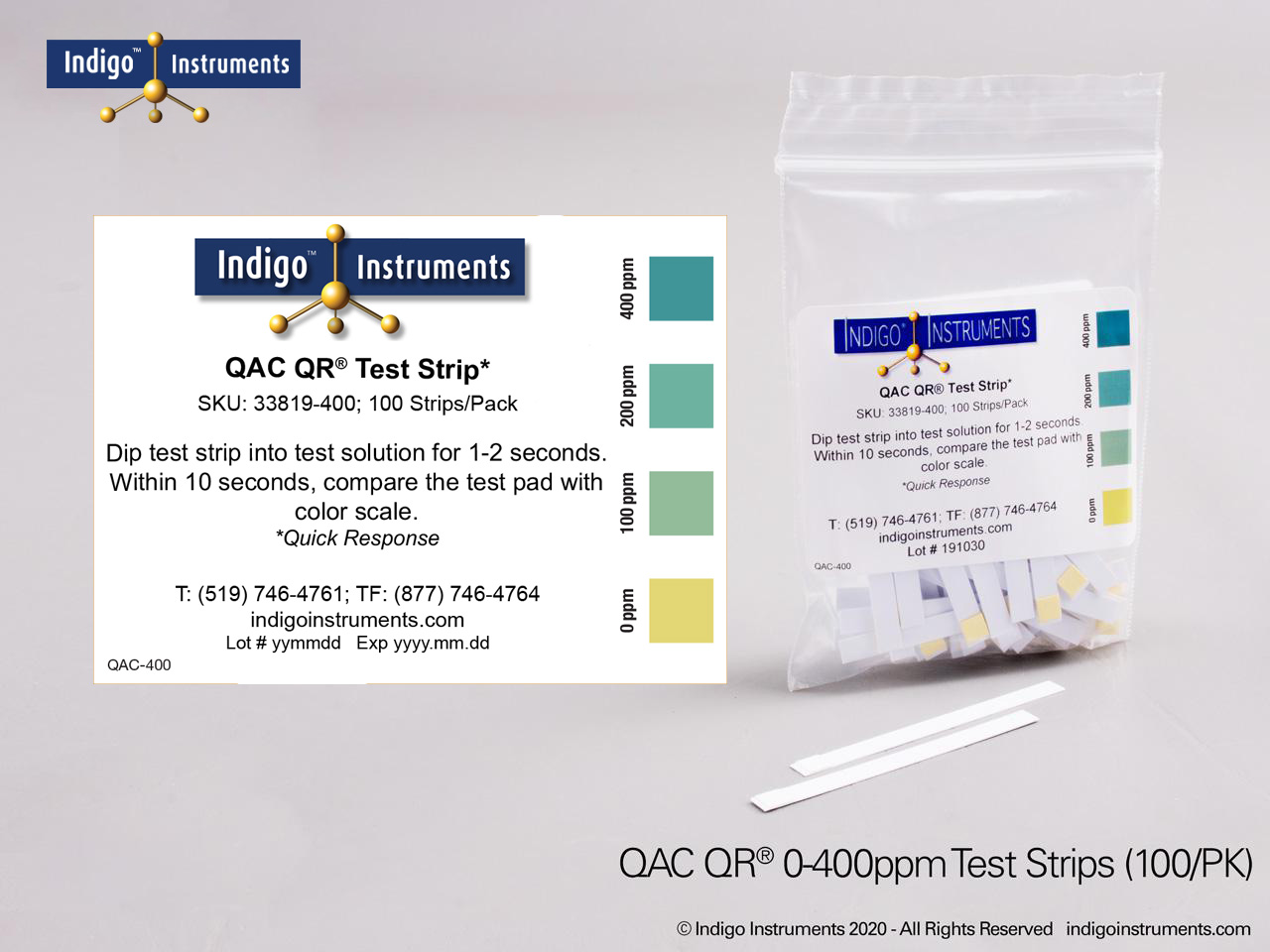
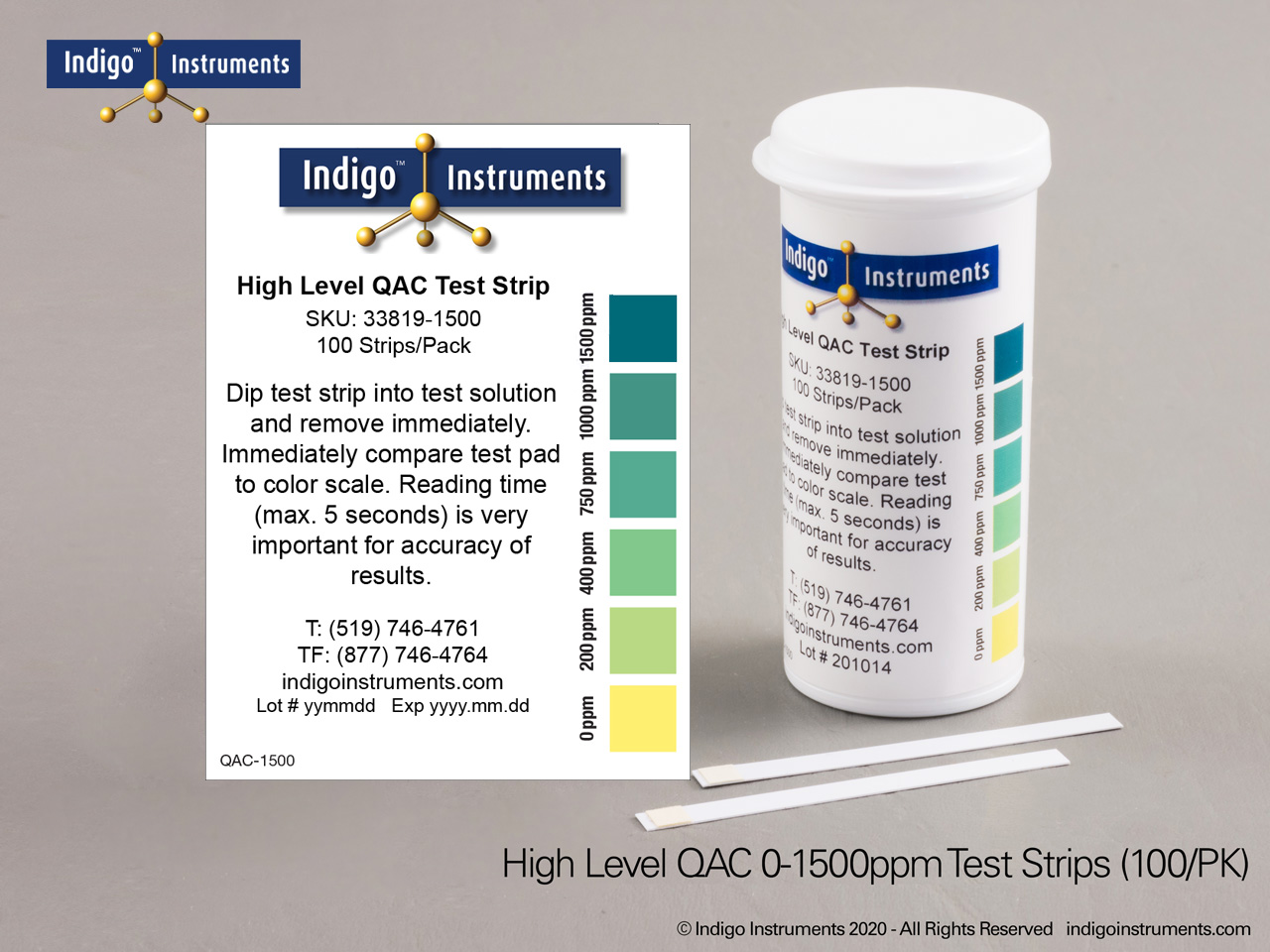
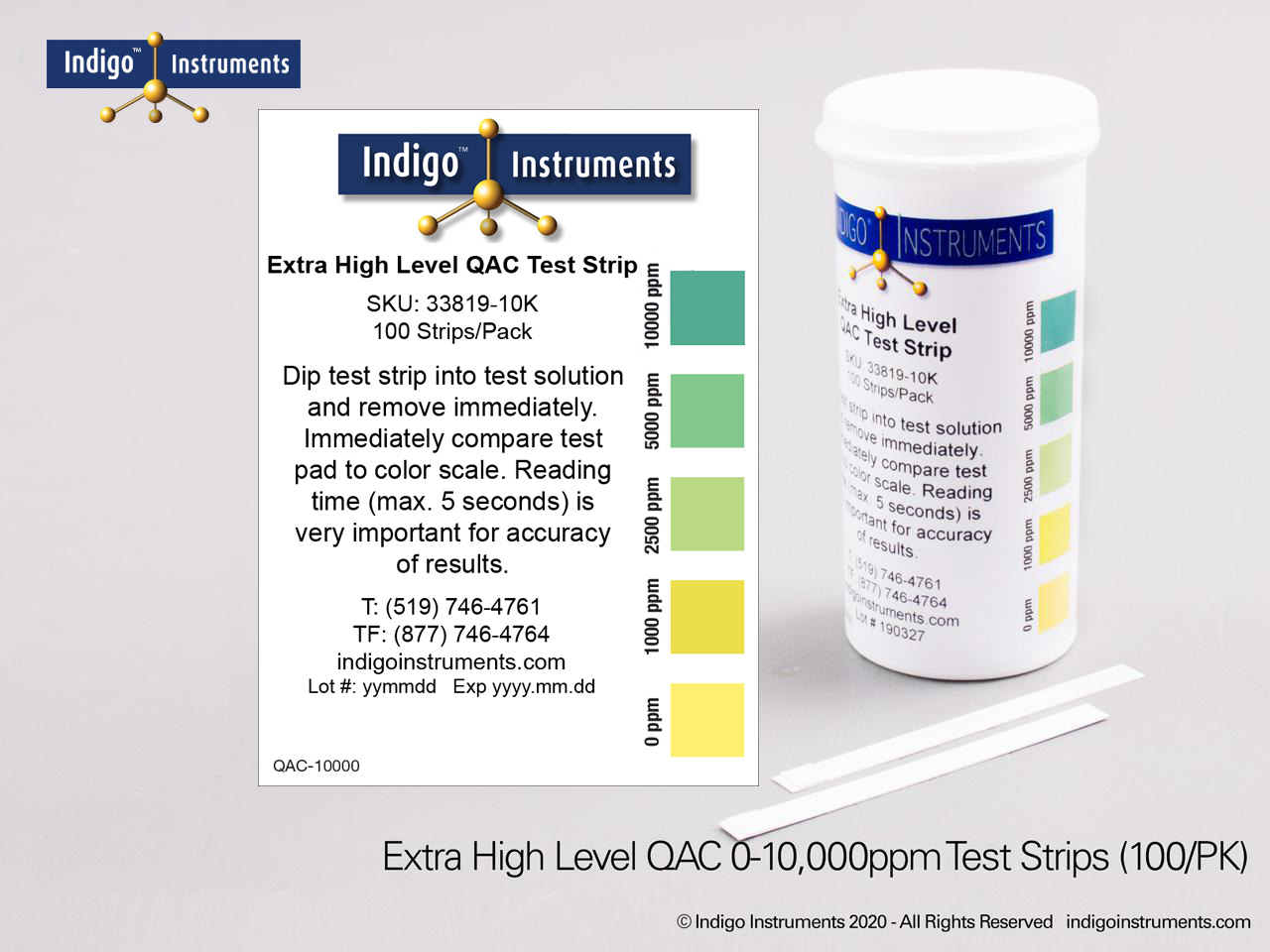
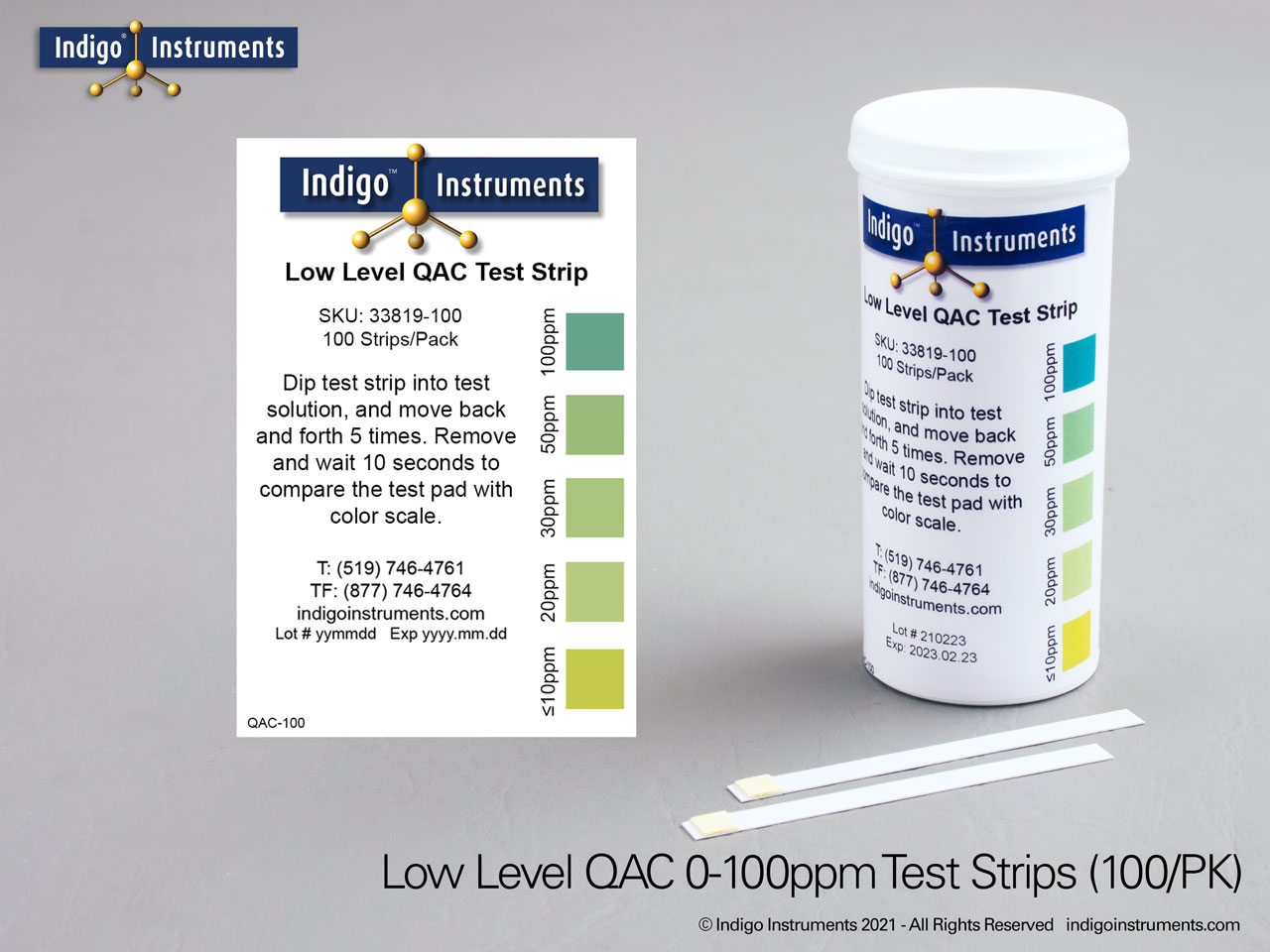
Indigo® Quat/QAC Test Strip Ranges
- 10-100ppm: ≤10-20-30-50-100ppm; read results in 10 seconds
- 0-400ppm: 0-100-200-300-400ppm; read results in 10 seconds
- 0-1500ppm: 0-200-400-750-1000ppm; read results in 5 seconds
- 0-10000ppm:0-1000-2500-5000-10000ppm; read results in 5 second
Common Features of Indigo® QAC Test Strips
- clear instructions with the labeled color chart
- indicators on high contrast plastic for accurate readings
- stiff plastic strips can be dipped into tubes
- accurate from 5-45oC (50-110oF)
- 3 year guaranteed shelf life (from date of manufacture shown as yyyy.mm.dd)
- Certificate of Analysis & Stability Statement, SDS online;
Certificate of Conformance on request.
Note: All test strips Made in the USA & are Q/A'd against NIST standards.
What our Customers Say
(Real Reviews-Not AI Generated Fakes; Click on the name to view the source at Google.)
Roberta Vigil: Indigo Instruments defines the purpose for the test strips and places them in easy to recognize categories. ...The BONUS is the qty of strips in each vial (100 vs the typical 50), fast and consistently accurate all for a price that beats other sellers!
Ronald Rosenberg: ...The sales person I spoke with was excellent and I received the items exactly when she said I would. Best of all the product was excellent. There was a clear delineation of the colors on the test strip so you could differentiate the different ppm. I would definitely repurchase this item.
Helen Kees: ...I loved the quality at a cost less than Amazon. But the real deal that drove me to leave a review was that I called back a week or so after receiving the product and asked for spec sheets requested by my organic inspector. I was treated like I had spent a million bucks and the info was at my fingertips within a minute. This kind of service is invaluable.