Glucose Enantiomers
SKU: 68845NV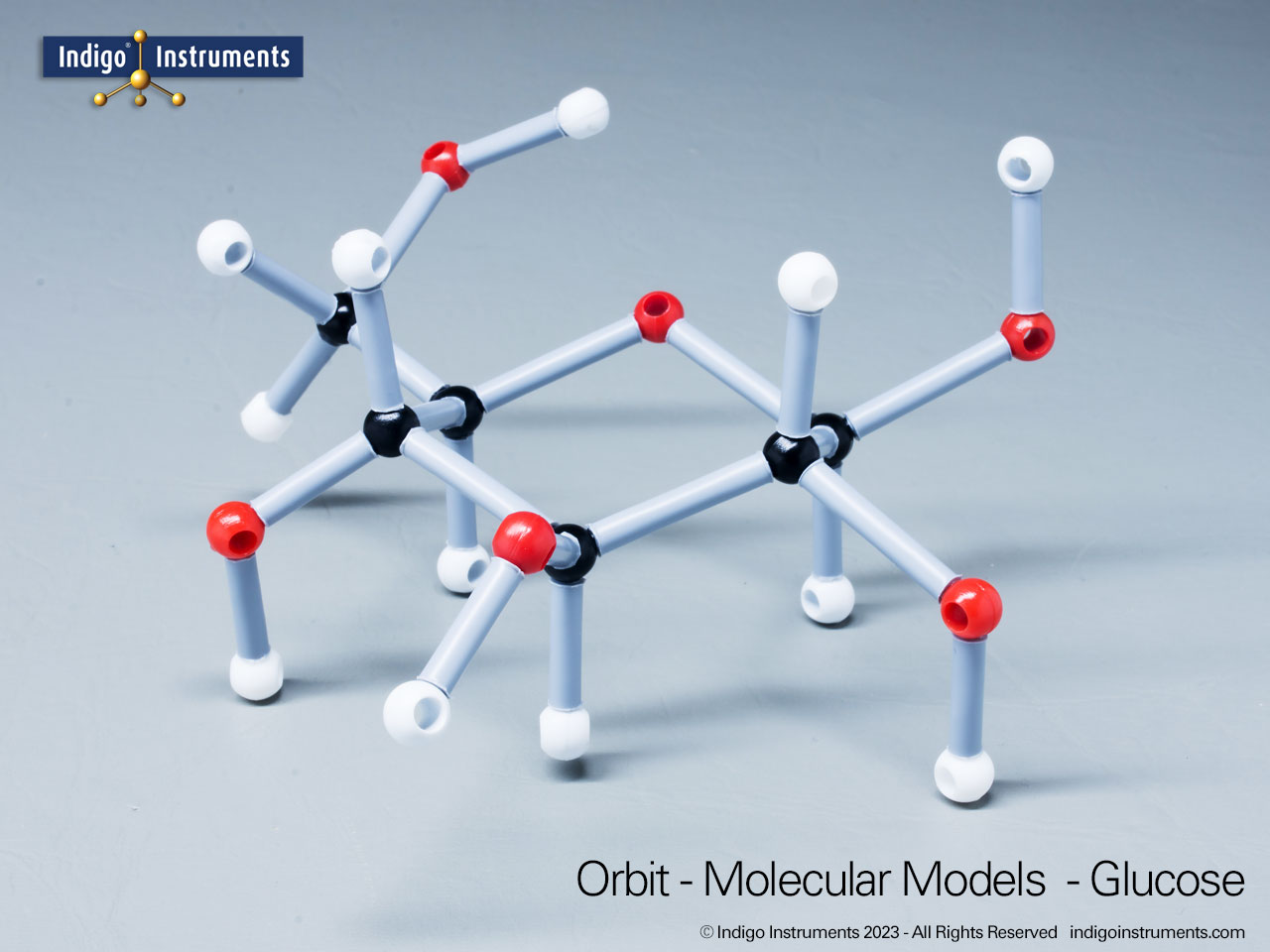
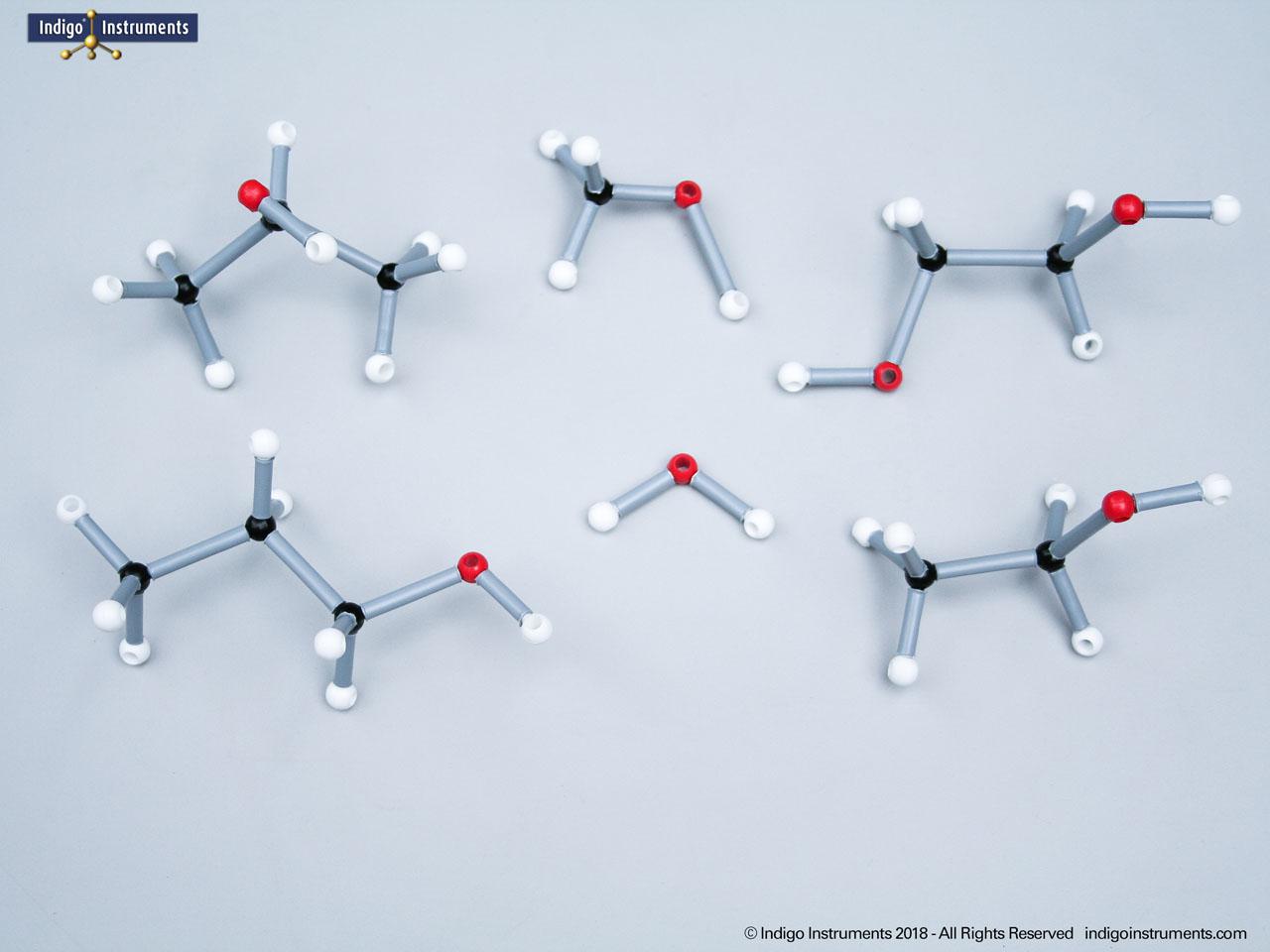
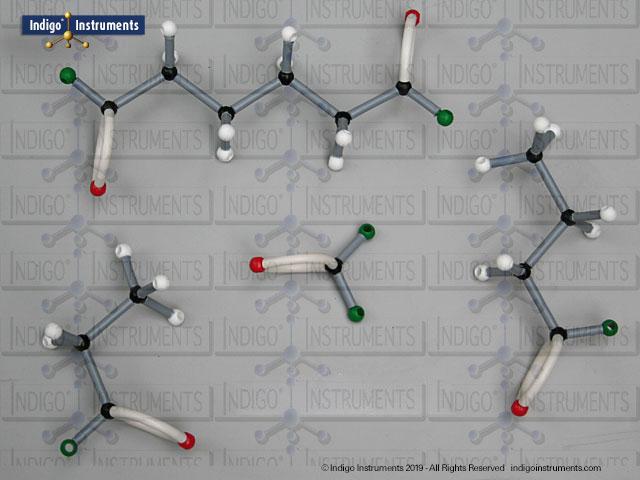
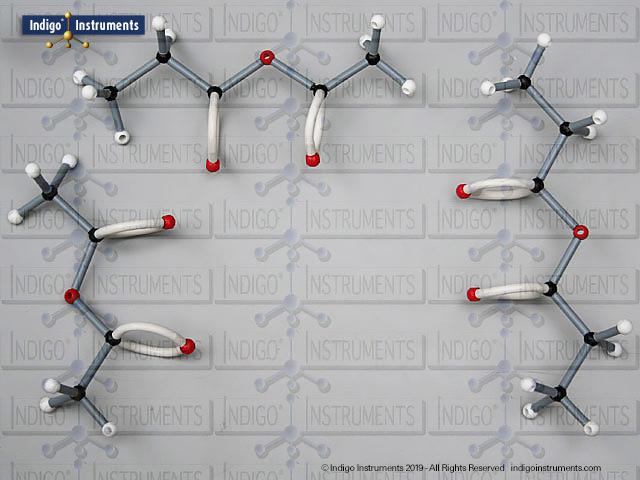
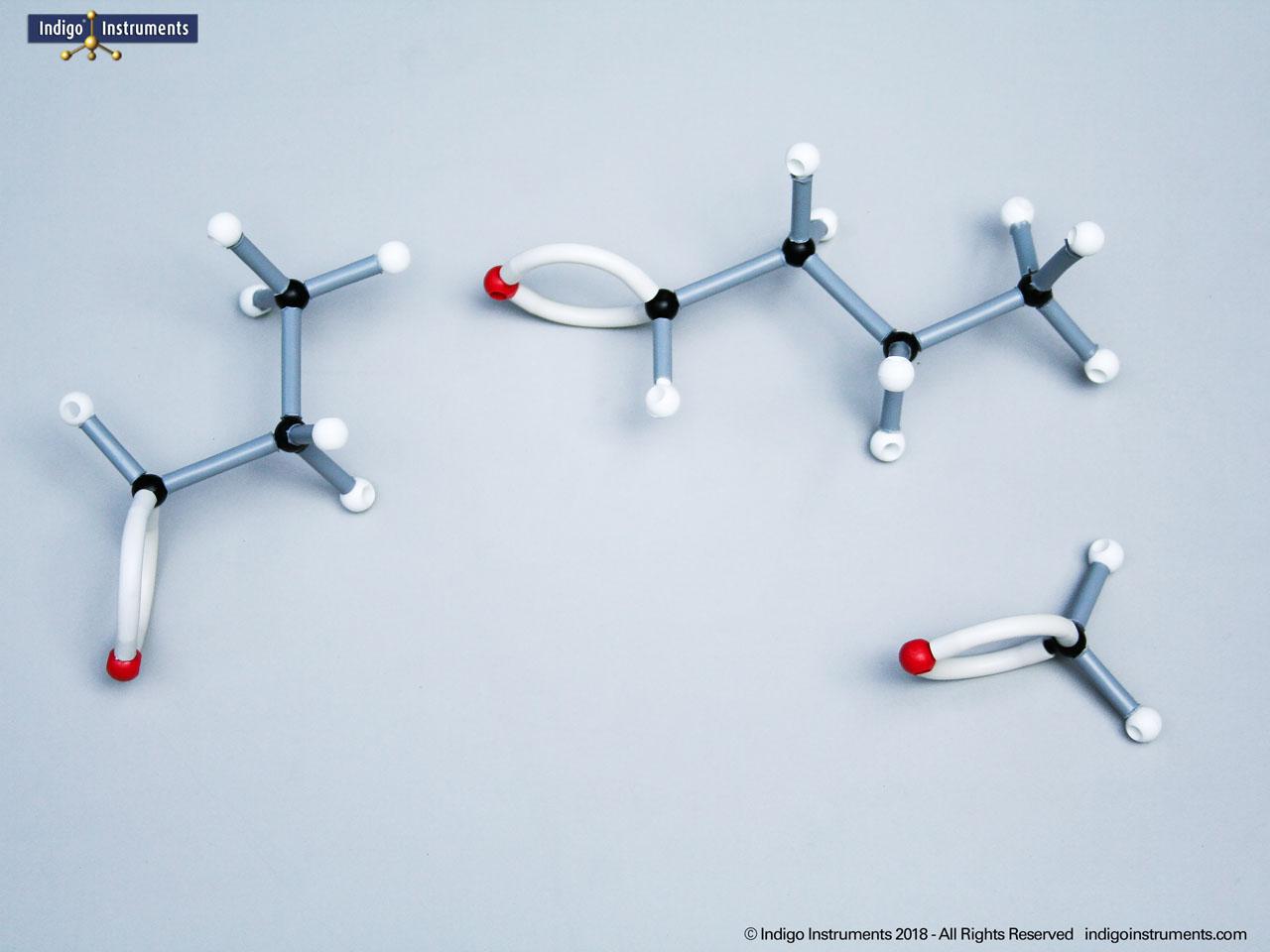
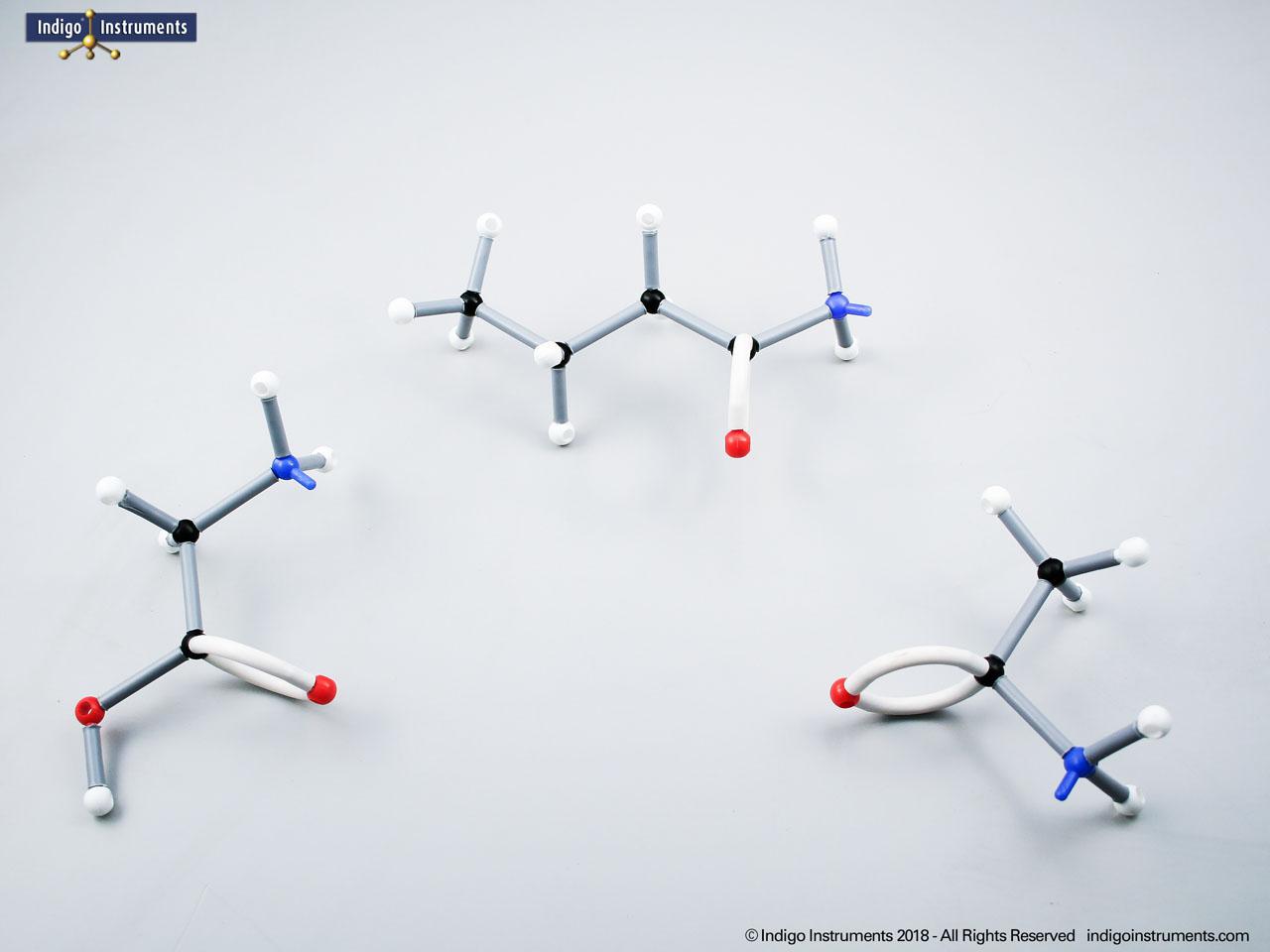
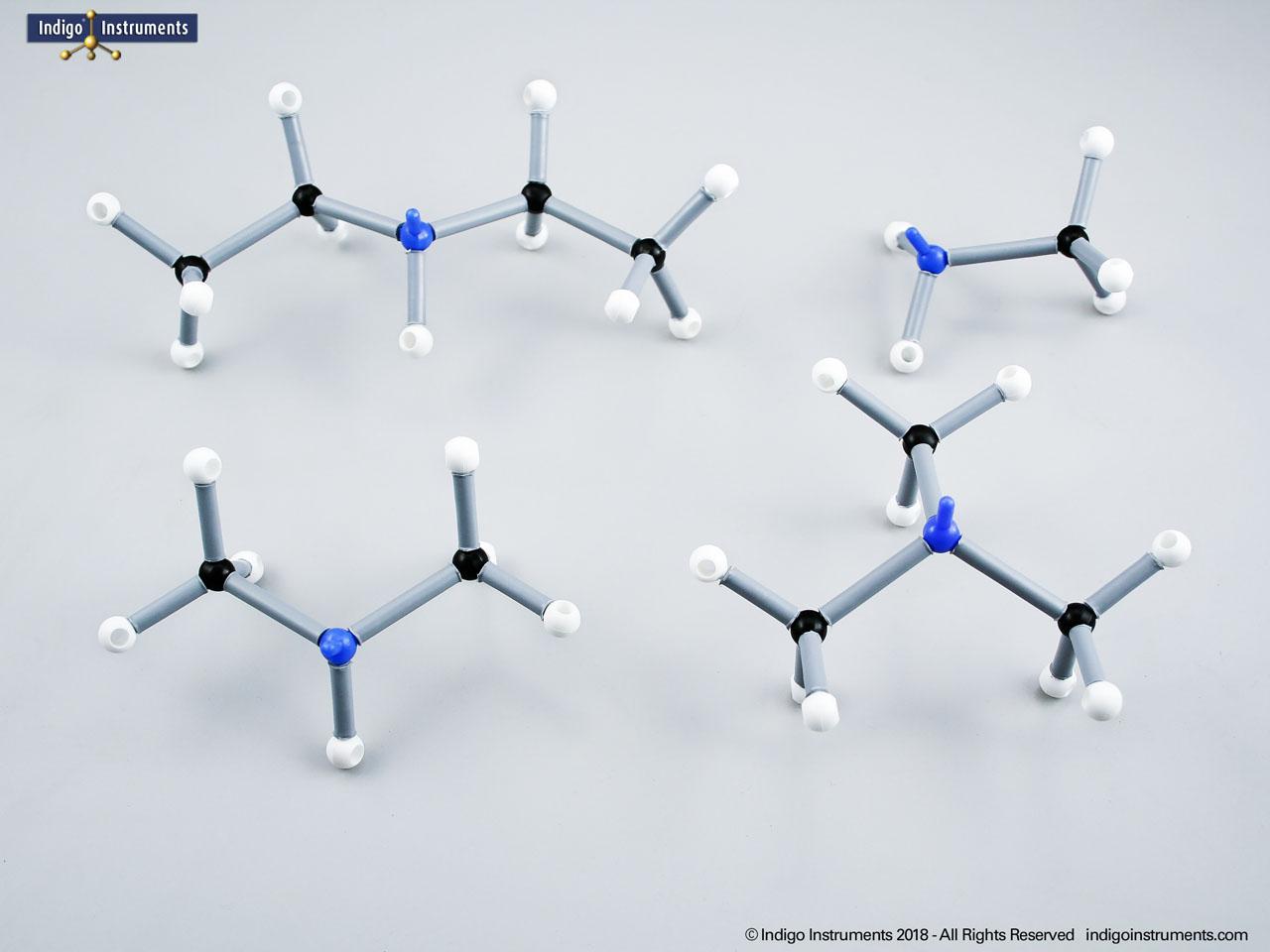
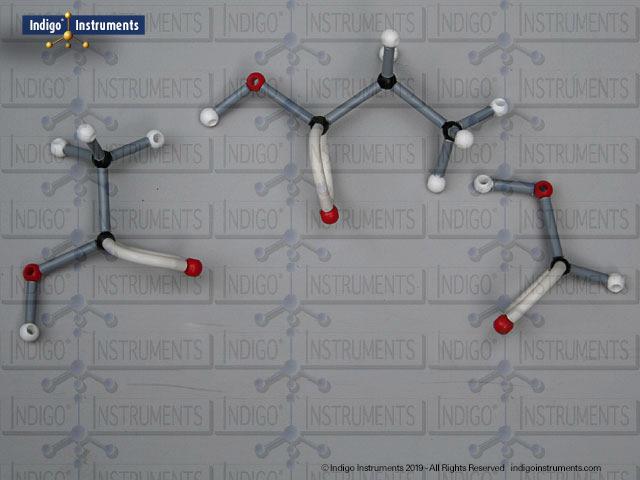
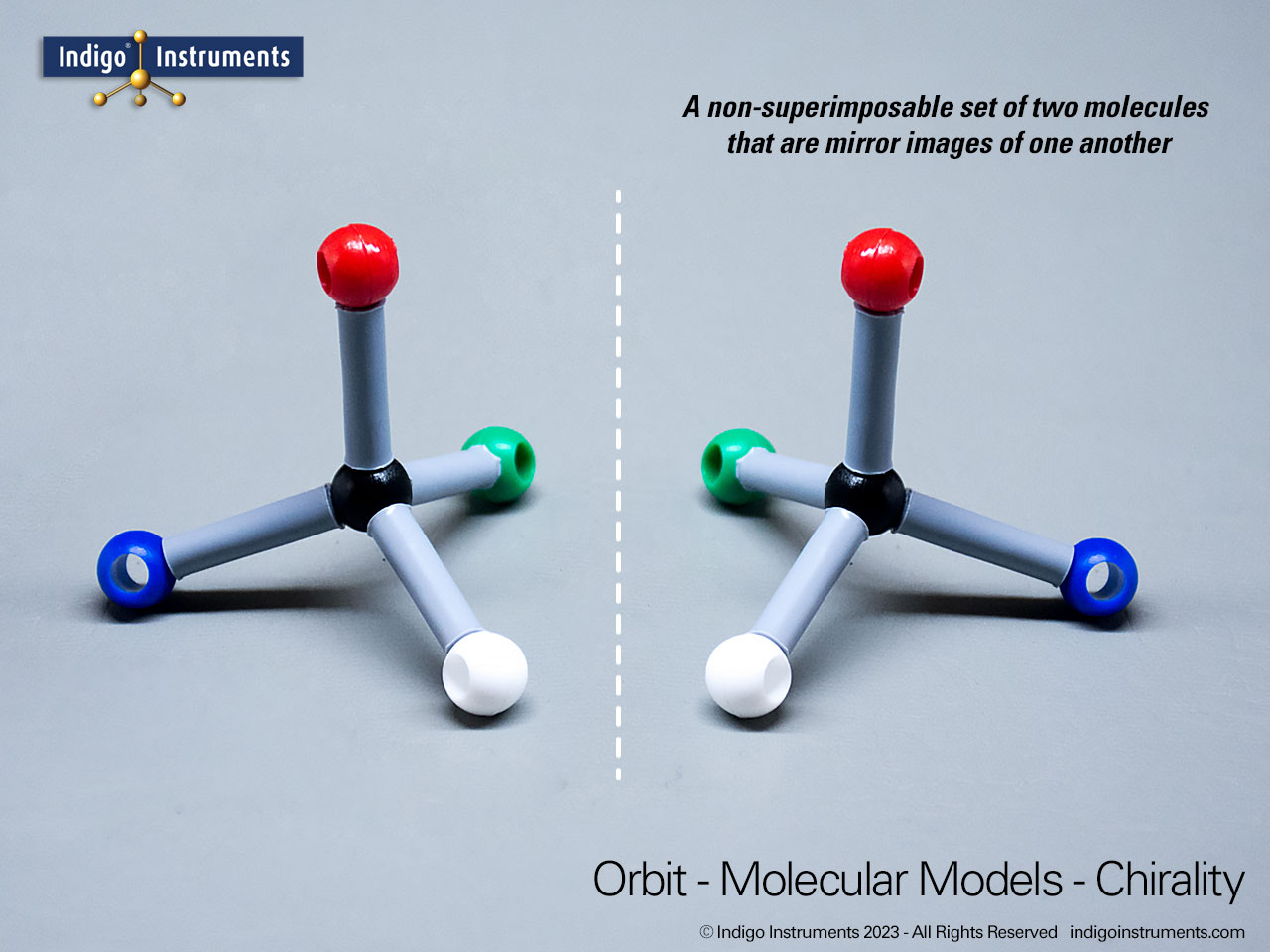
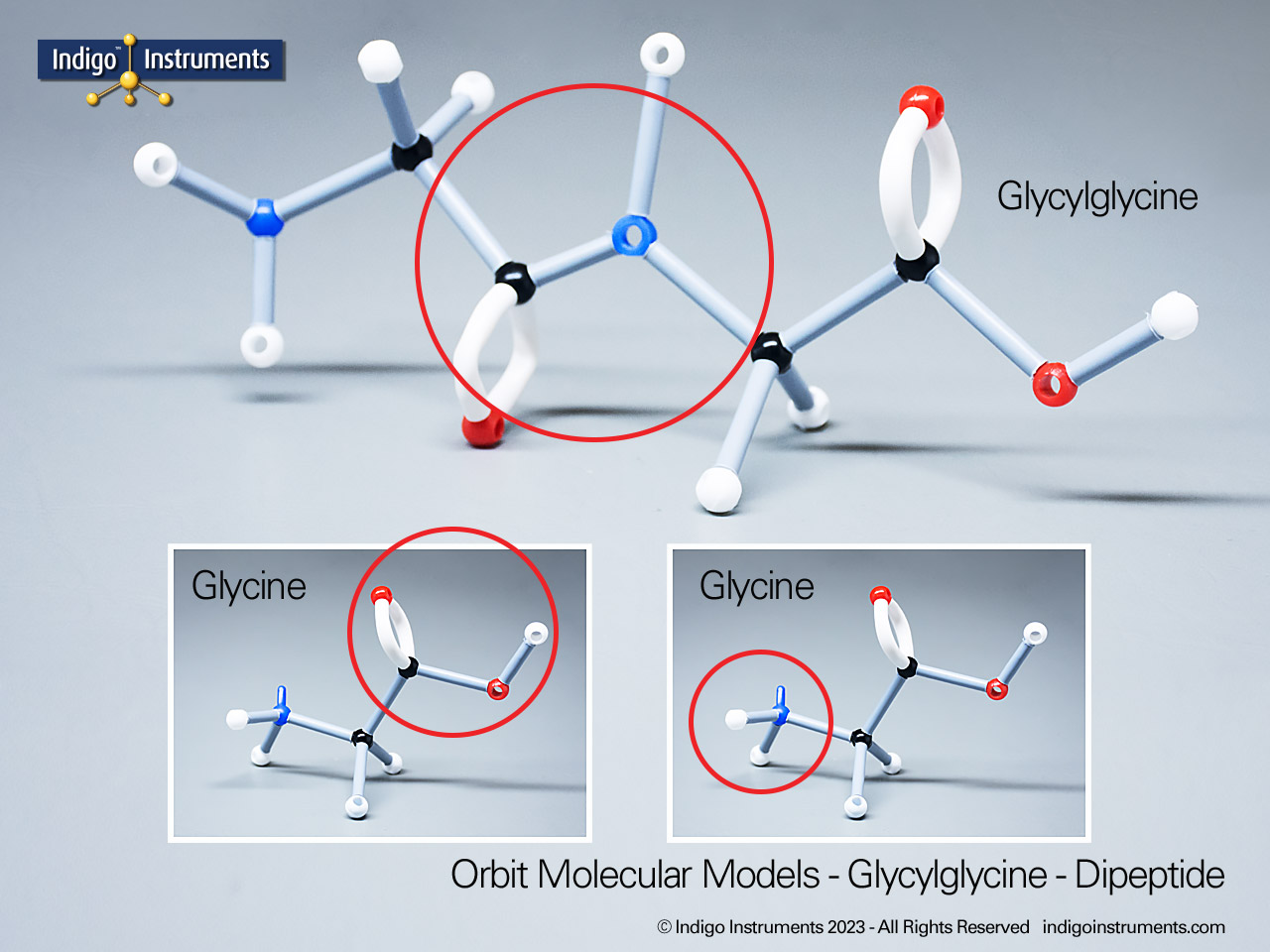
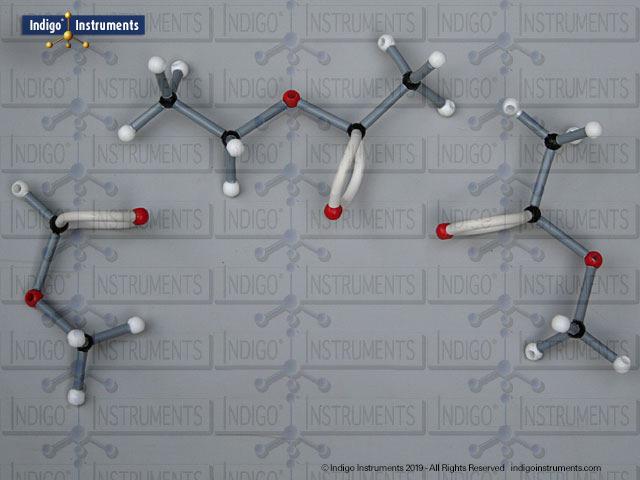
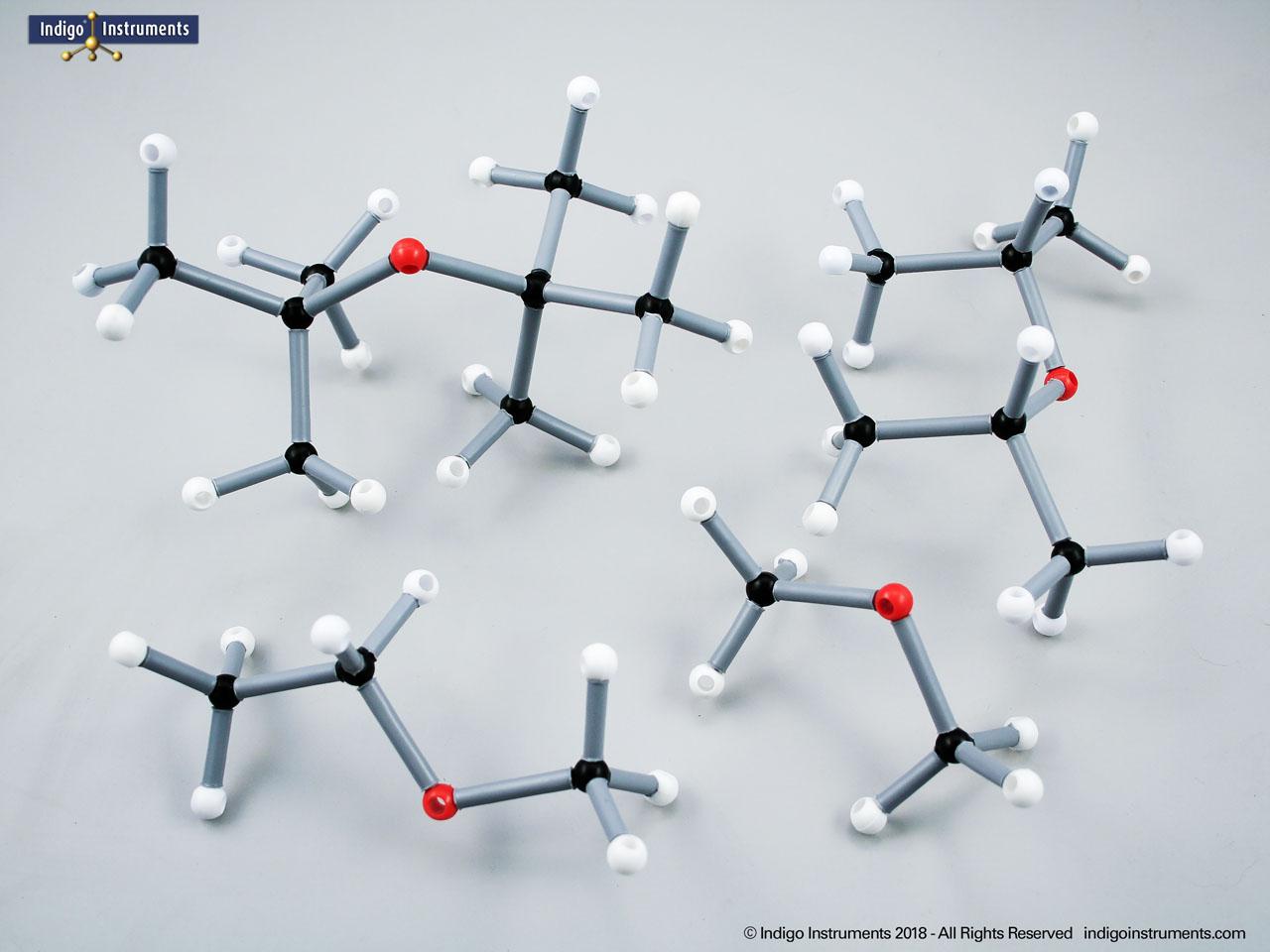
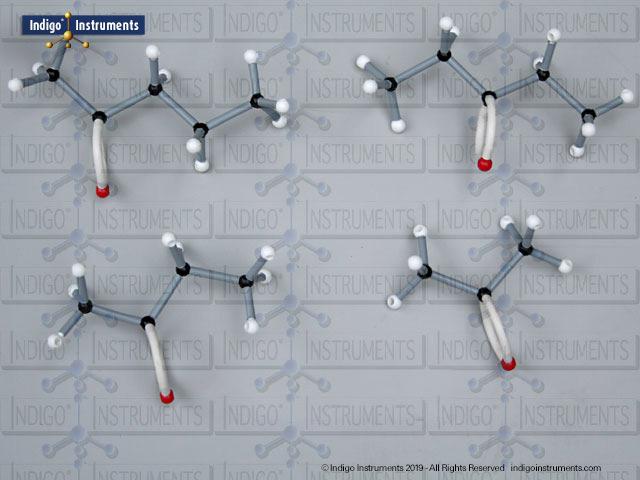
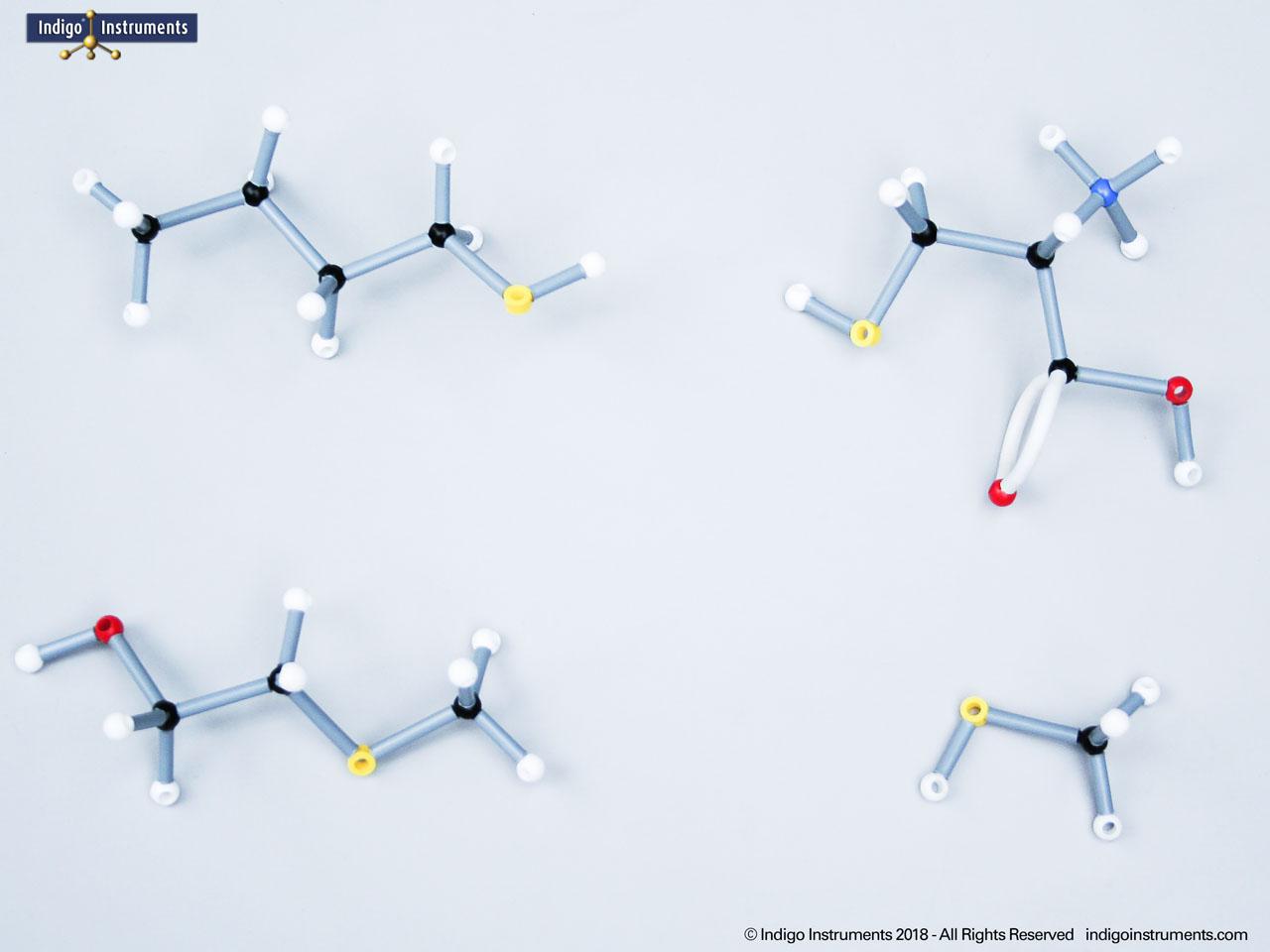
Building models of glucose isomers is a practical way to understand stereochemistry. Glucose has 16 possible stereoisomers in linear form & 32 ring forms. These include galactose, maltose & others, all of which can take on chair & boat conformations similar to cyclohexane. This is especially of interest to biologists/biochemists since it relates chemical structure to enzyme specificity.
Building physical models of glucose and its isomers using organic chemistry molecular model kits is one of the most effective ways to visualize stereochemistry, especially for students preparing for the MCAT or enrolled in introductory biochemistry. In its linear form, glucose has 16 stereoisomers, and its ring forms can number up to 32, including biologically significant sugars such as galactose, mannose, and maltose.
These ring structures adopt chair and boat conformations, just like cyclohexane, making model-building kits with stiff bonds such as this ideal for accurate ring flipping. Understanding these 3D shapes is critical to grasping how enzyme-substrate specificity works, particularly in processes like glycolysis, glycogen storage, and cell surface recognition.
Indigo Instruments has stocked genuine Cochranes of Oxford molecular model parts for 30+ years. Customize or replenish any model set/kit we have ever sold!