Alkane Chemical Structure Models
SKU: 68845NV
Discover alkane molecular models for teaching and research. Learn how straight-chain, branched, and cyclic alkanes illustrate key concepts in organic chemistry, biochemistry, and MCAT preparation.
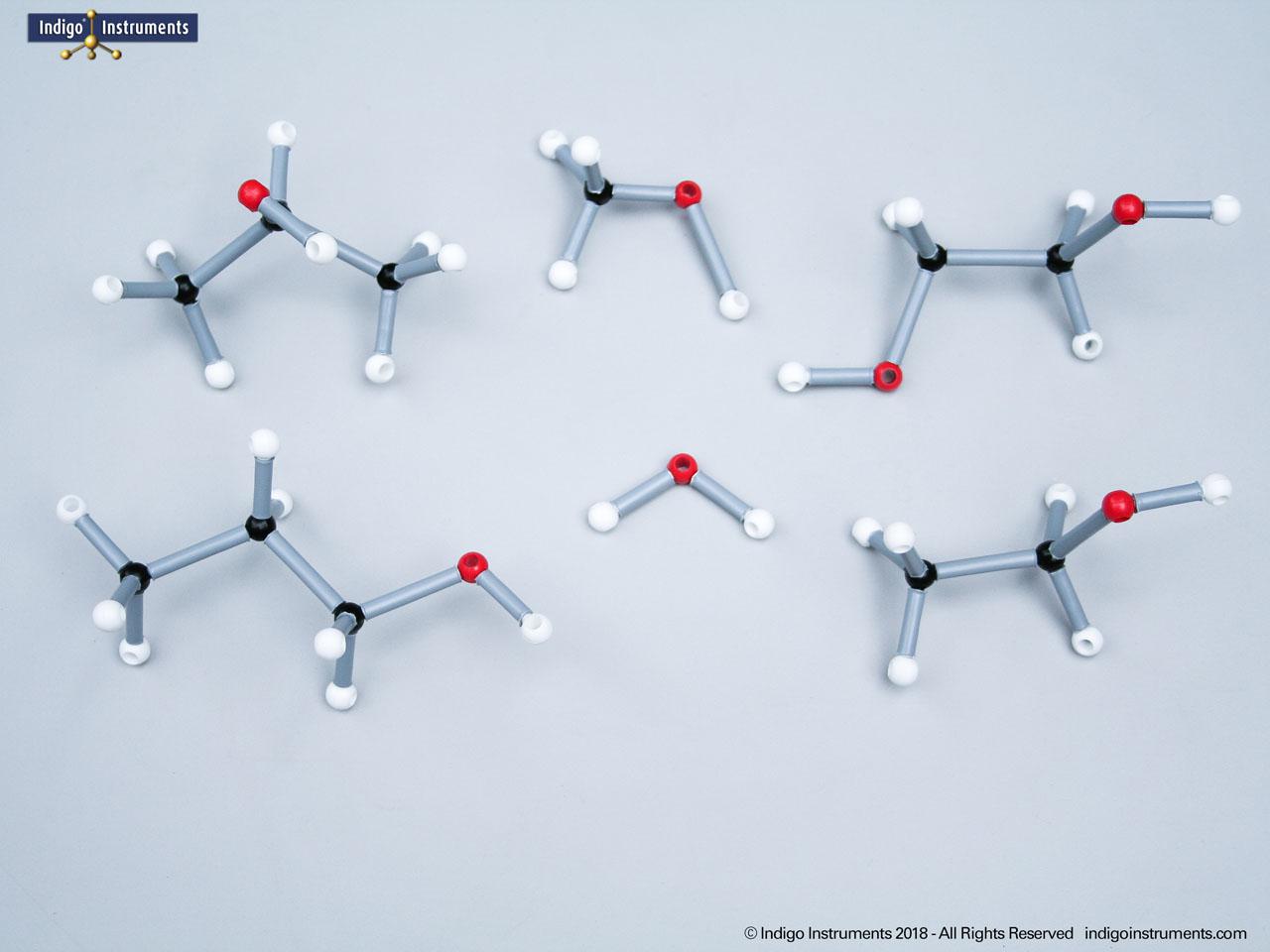
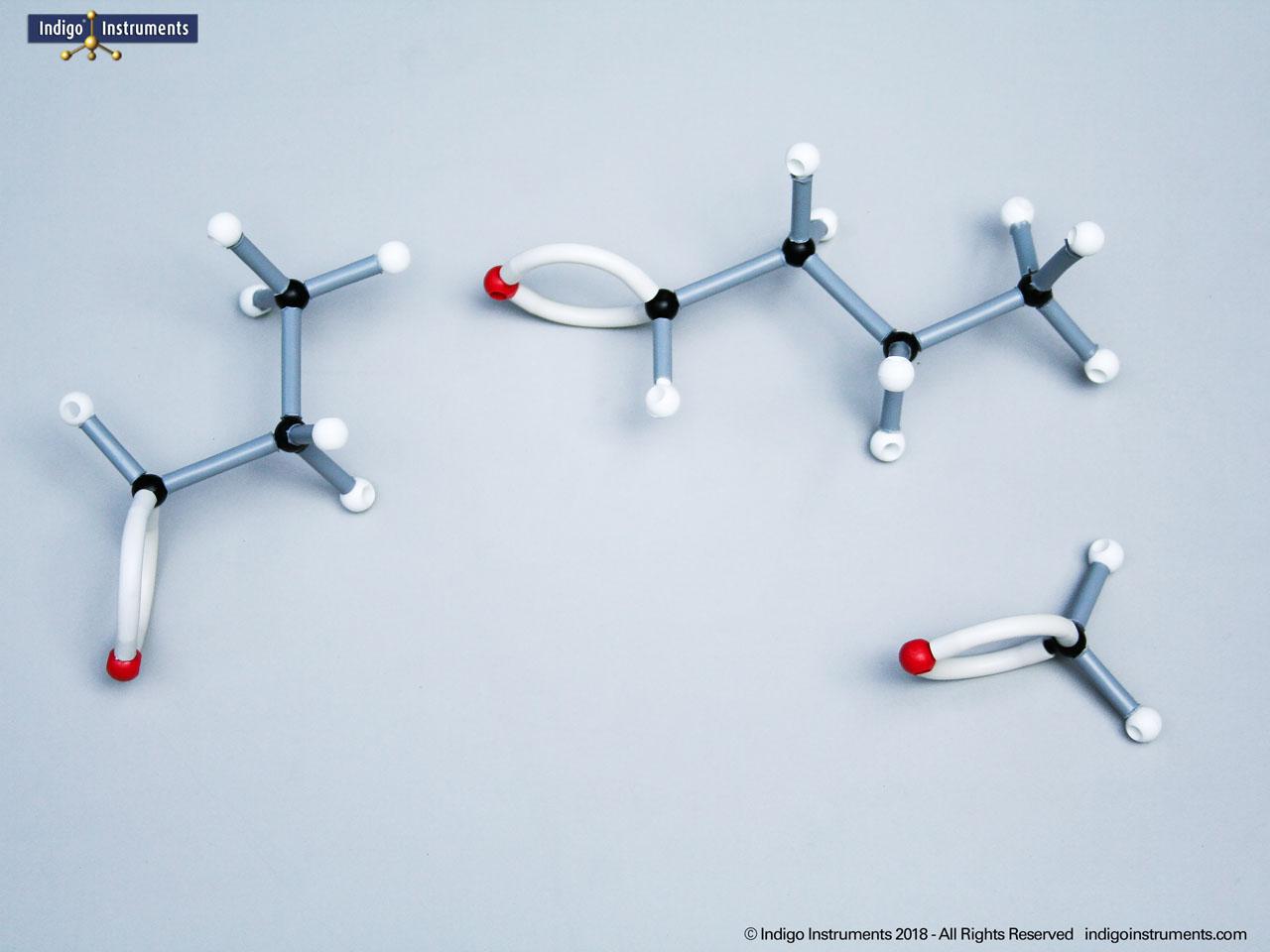
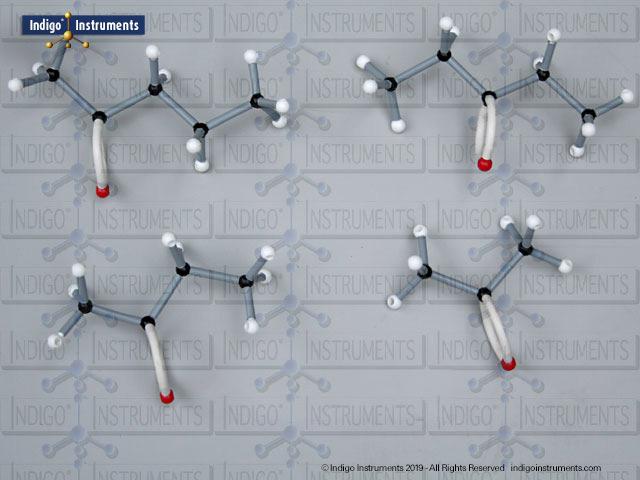
Alkanes are the simplest organic compounds. They are nonpolar & water insoluble (hydrophobic) with different physical and chemical properties. These saturated hydrocarbons are fundamental to understanding organic chemistry. Alkane molecular models clearly demonstrate the tetrahedral geometry of carbon atoms, the effect of branching on physical properties, and the basis for IUPAC naming. In biochemistry and MCAT preparation, alkanes are an essential starting point for learning about hydrocarbon reactivity, isomerism, and the structural backbone of more complex biomolecules.
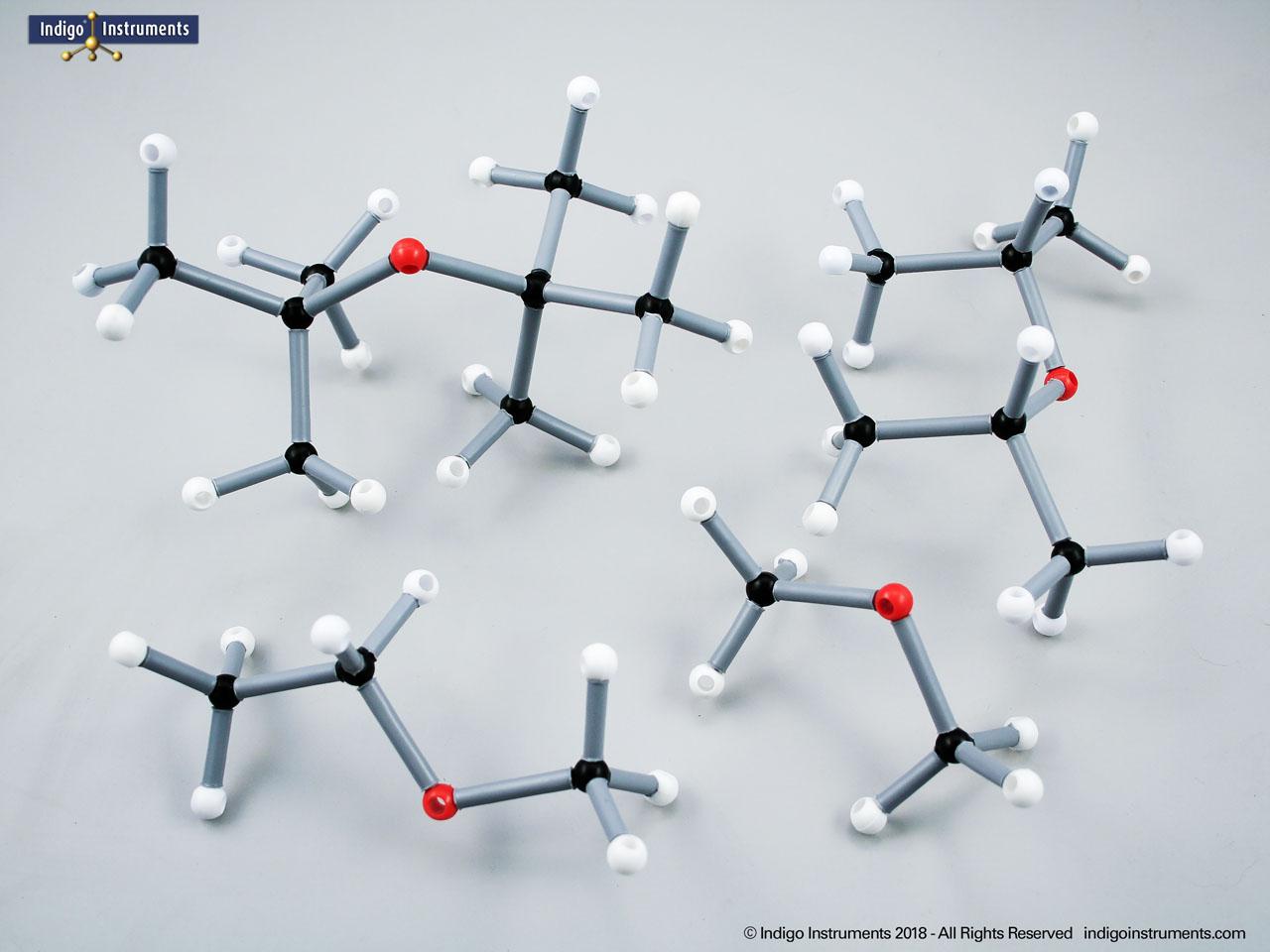
The 68845NV Foundation basic student molecular set can build standard straight chain alkanes such as CH4 (methane), C2H6 (ethane), C3H8 (propane), C4H10 (butane), C5H12 (pentane), C6H14 (hexane), C7H16 (heptane), & C8H18 (octane). Or, compare these 5 structural isomers: Hexane; 2-methylpentane; 3-methylpentane; 2,3-dimethylbutane; 2,2-diemethylbutane isomers.
Indigo Instruments has stocked genuine Cochranes of Oxford molecular model parts for 30+ years. Customize or replenish any model set/kit we have ever sold!