Acid Halide Functional Group Models
SKU: 68845NV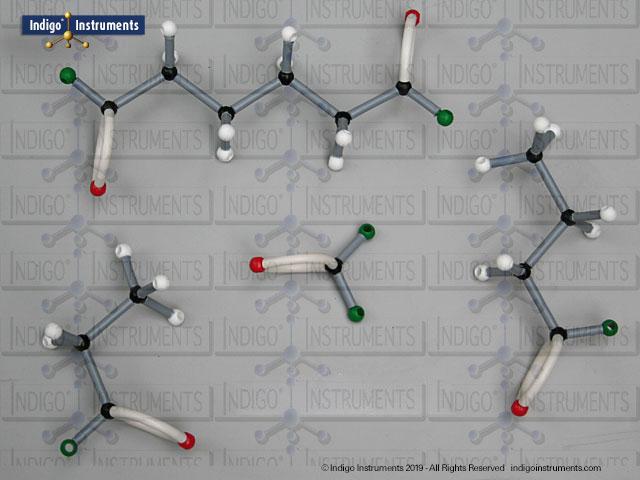
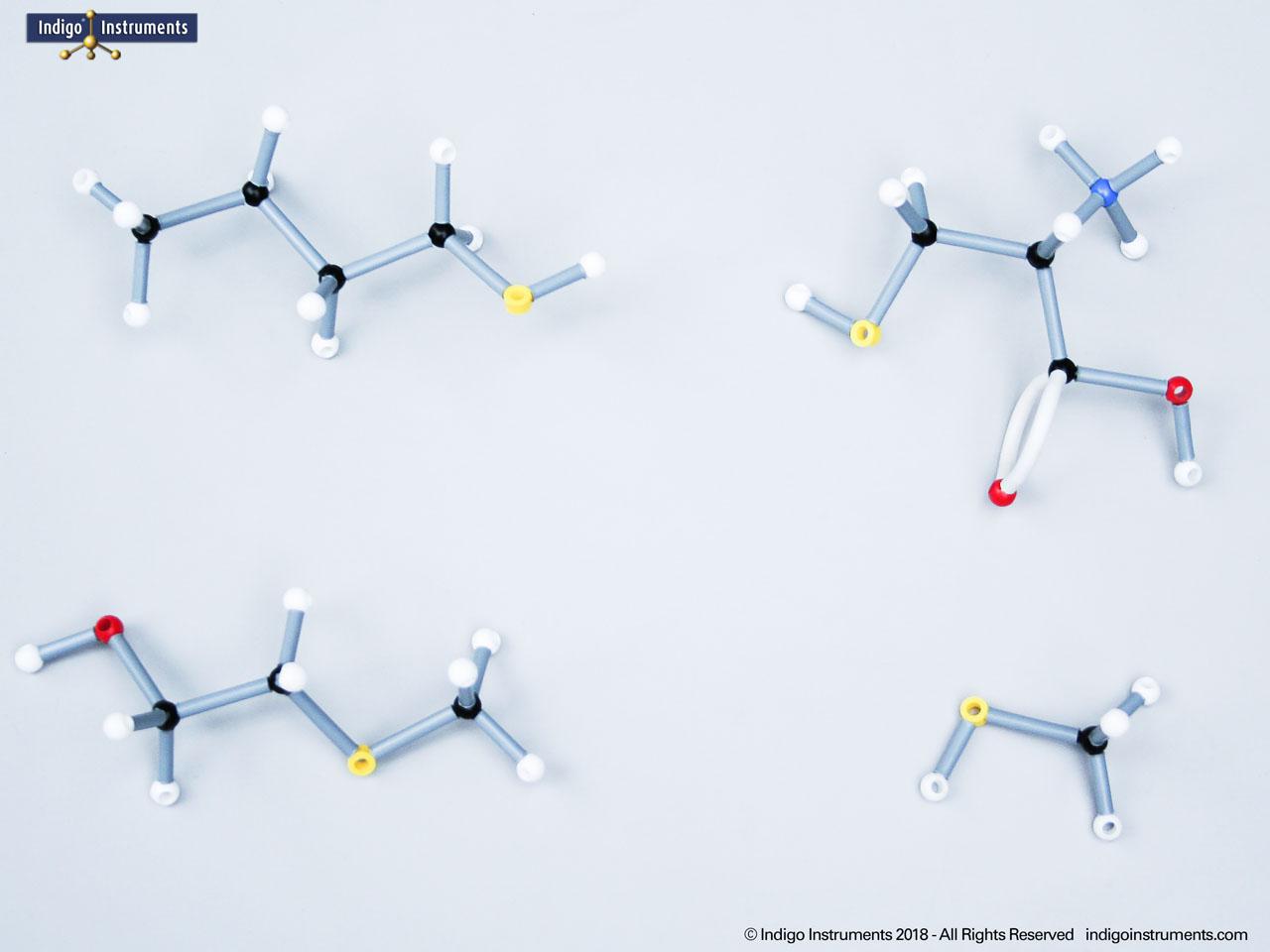
Build & compare acid halide molecular model structure for insights into chemical & biological applications. Learn about reactivity, acyl substitution, IUPAC naming, and relevance using Orbit models for hands-on visualization.
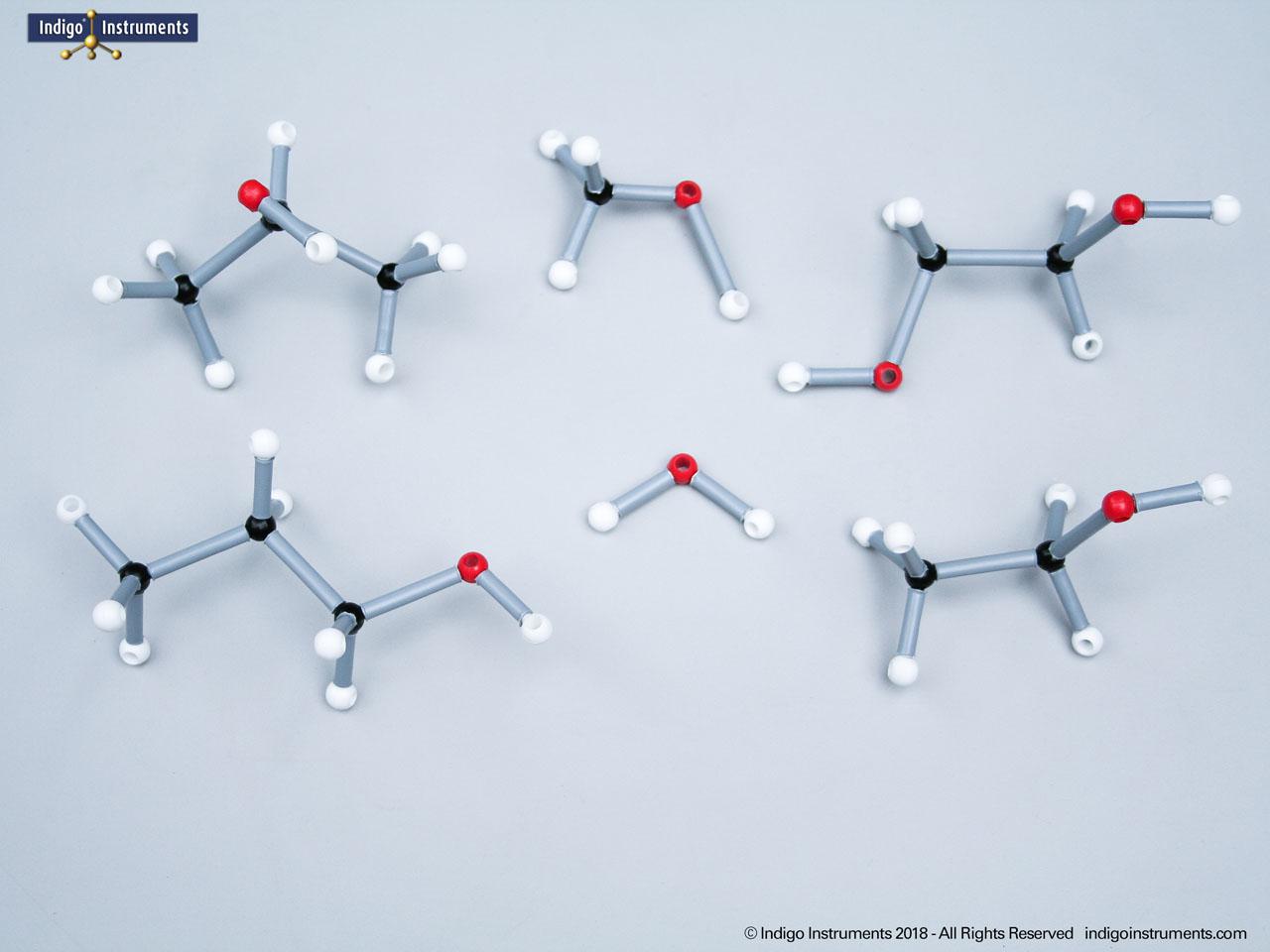
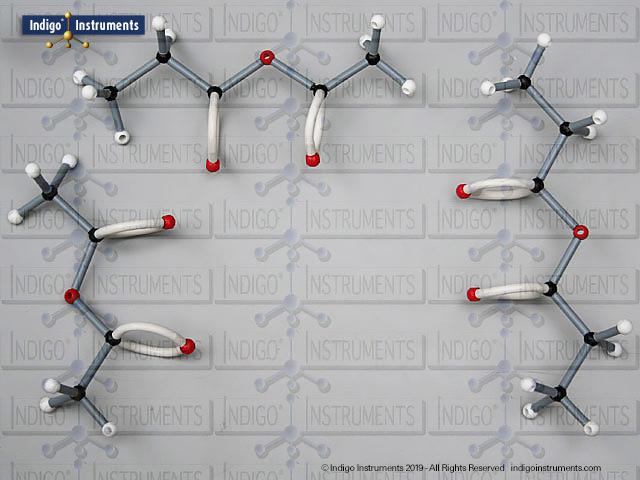
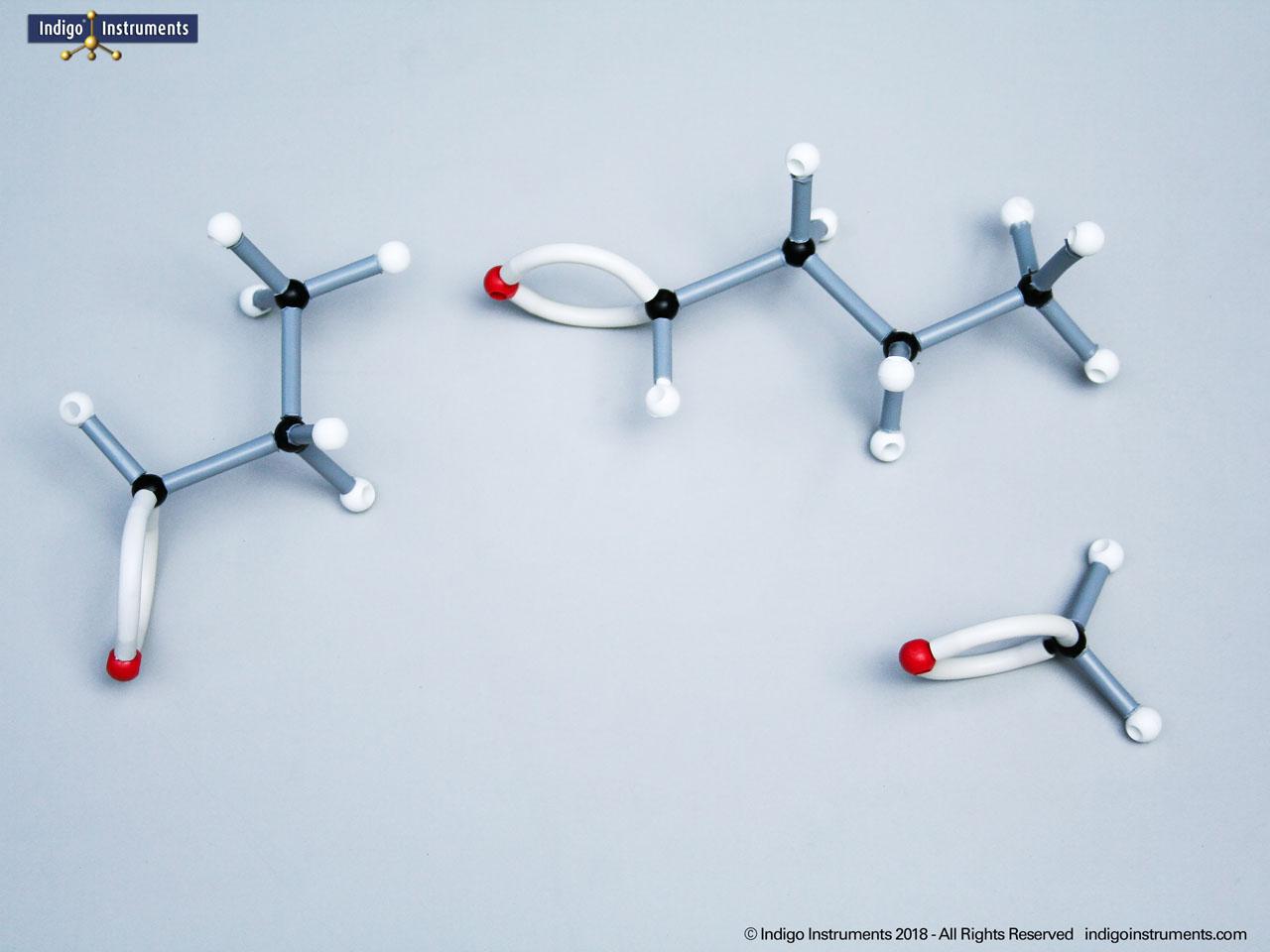
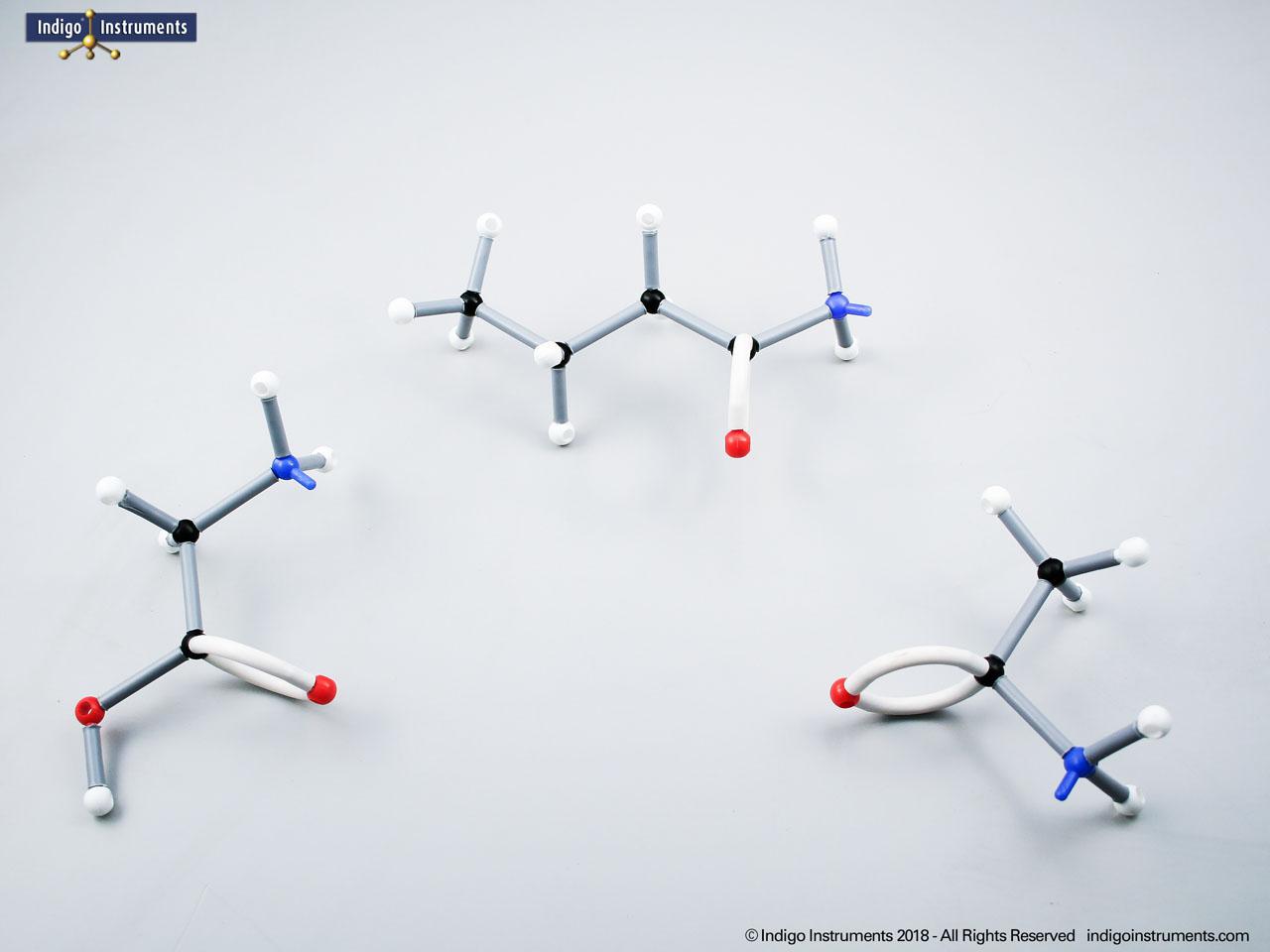
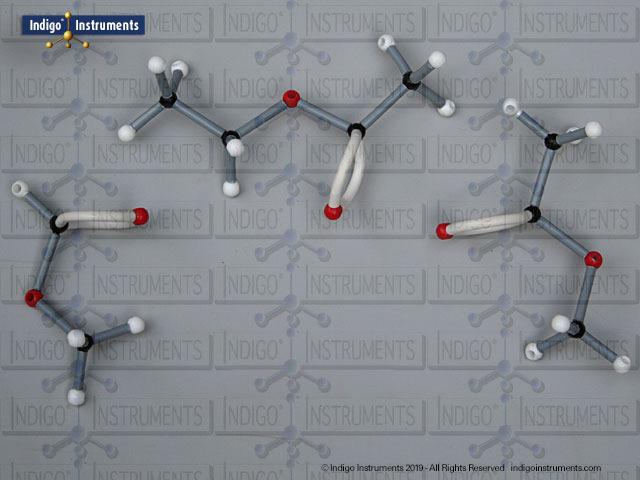
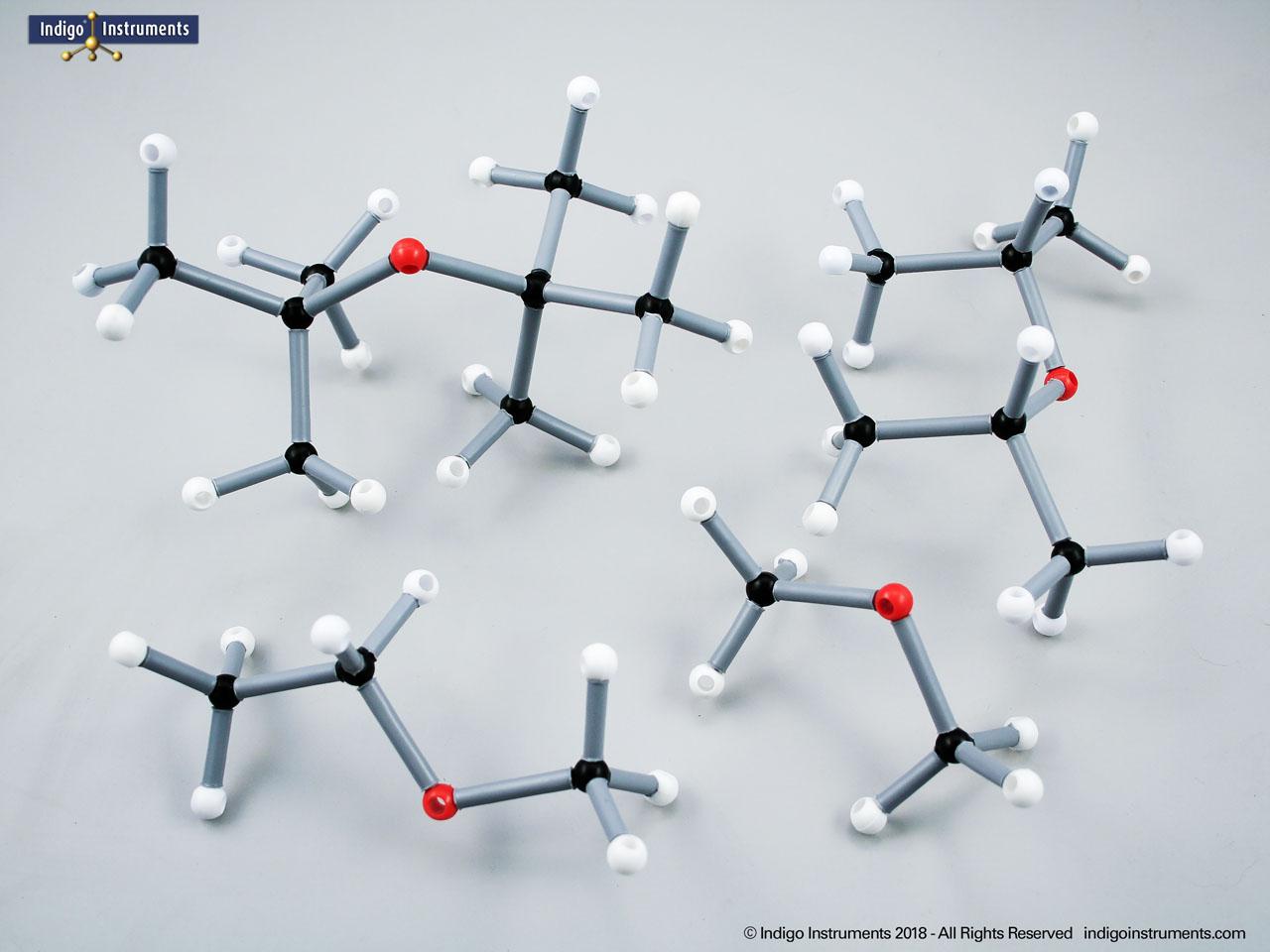
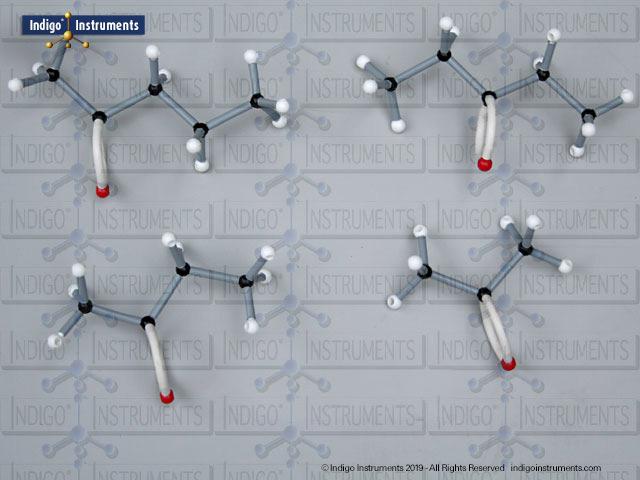
Acid halides (acyl halides) are highly reactive derivatives of carboxylic acids in which the hydroxyl group is replaced by a halogen atom, typically chlorine. They are essential in organic chemistry for studying acyl substitution reaction mechanisms, synthesis of esters, amides, and anhydrides. In biological and medicinal contexts, acid halide analogs can help illustrate enzyme-mediated acylation, drug synthesis intermediates, and chemical modifications. Orbit acid halide molecular models allow students to visualize the planar carbonyl carbon, bond angles, and the effects of halogen substitution. They are a versatile tool for chemistry & biology related majors.
Indigo Instruments has stocked genuine Cochranes of Oxford molecular model parts for 30+ years. Customize or replenish any model set/kit we have ever sold!