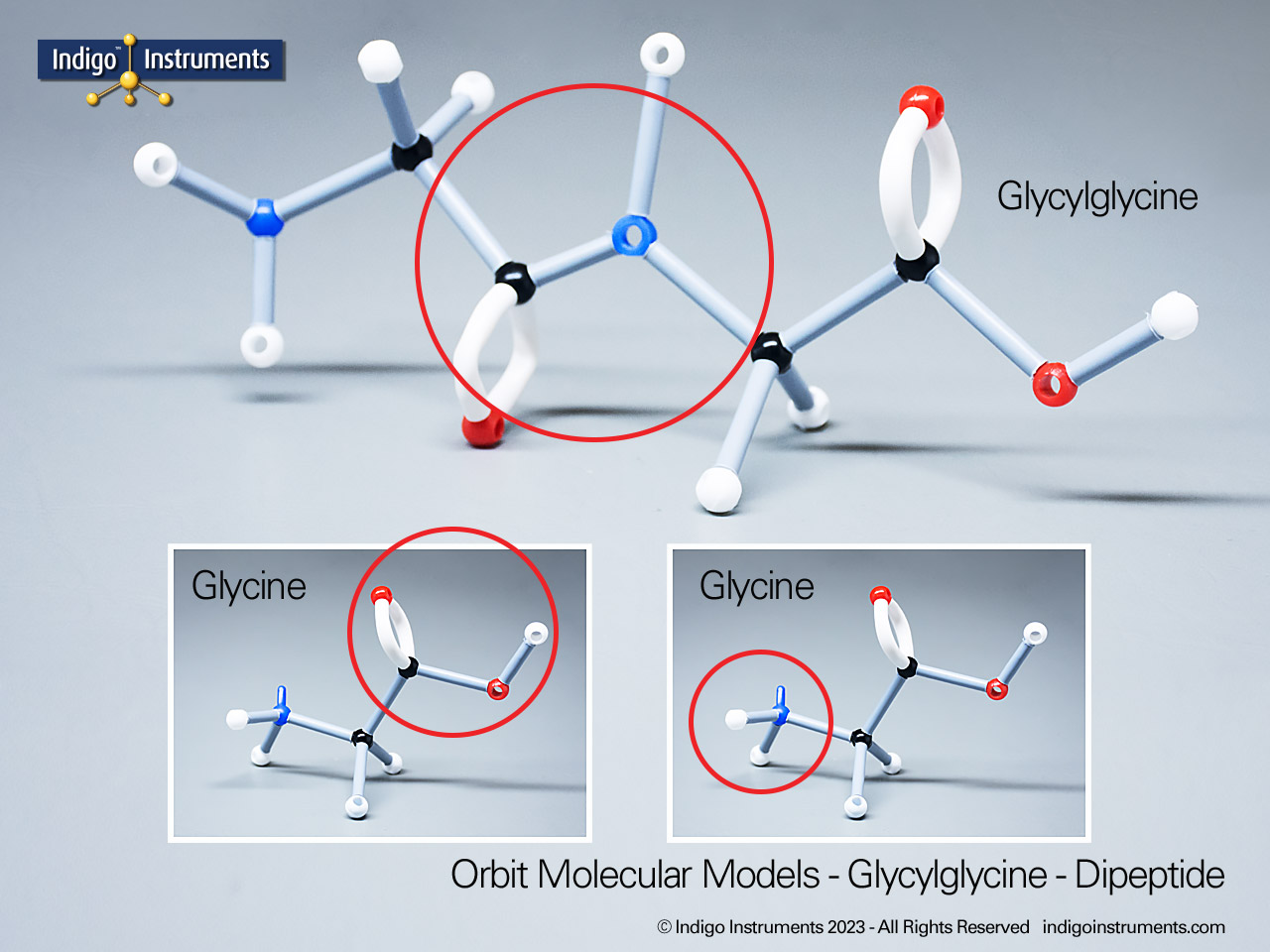
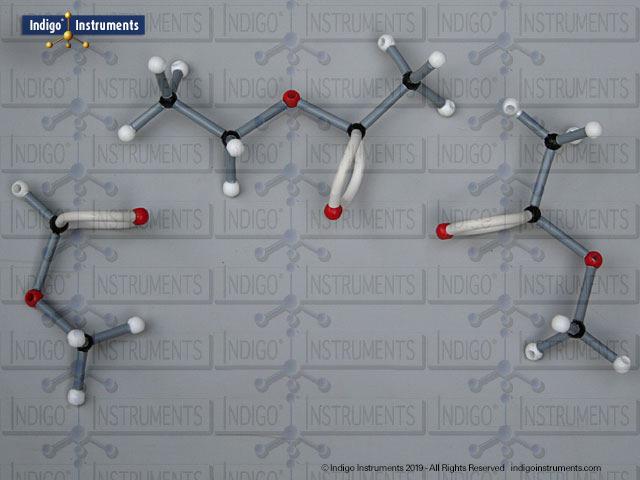
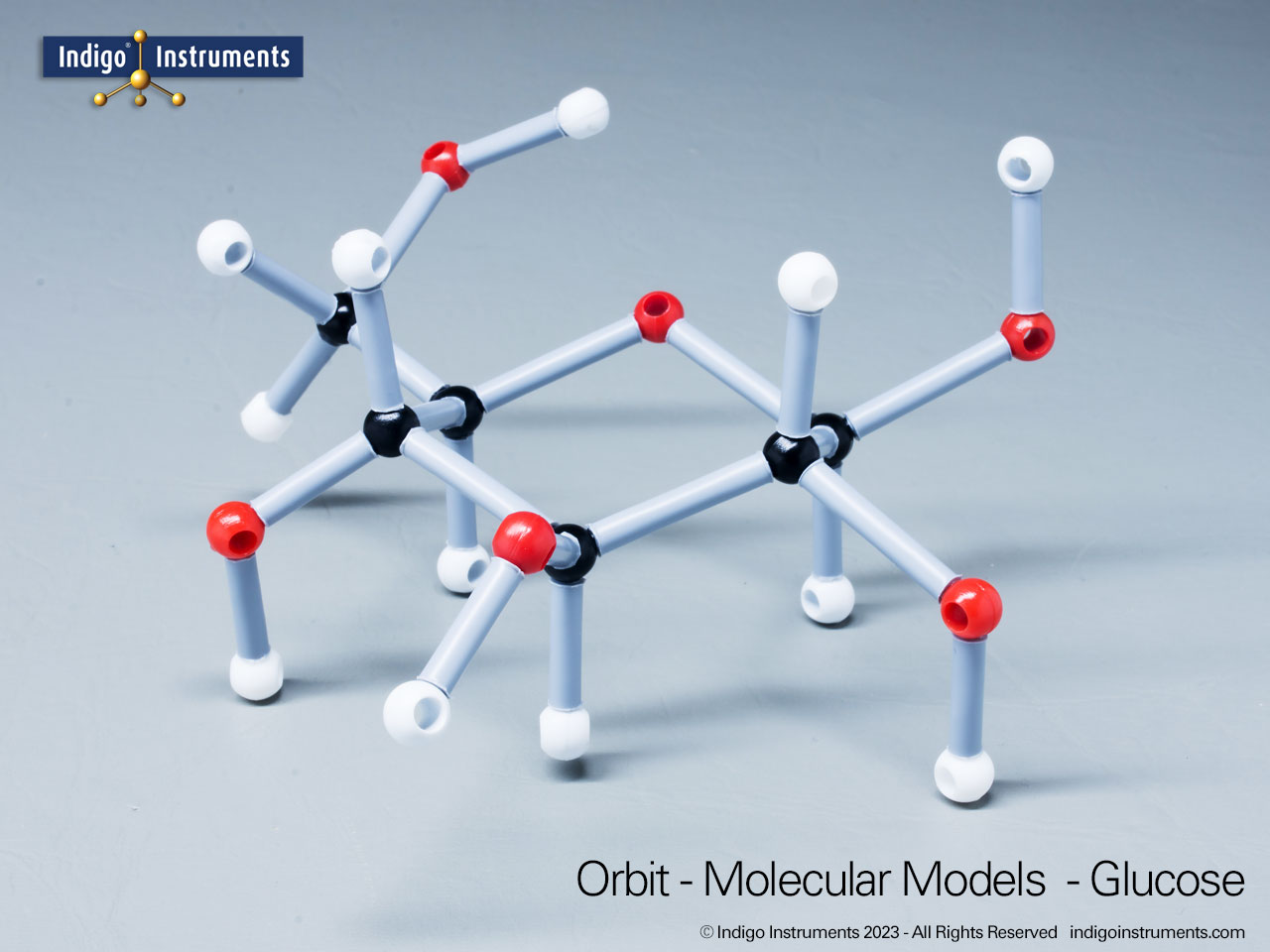
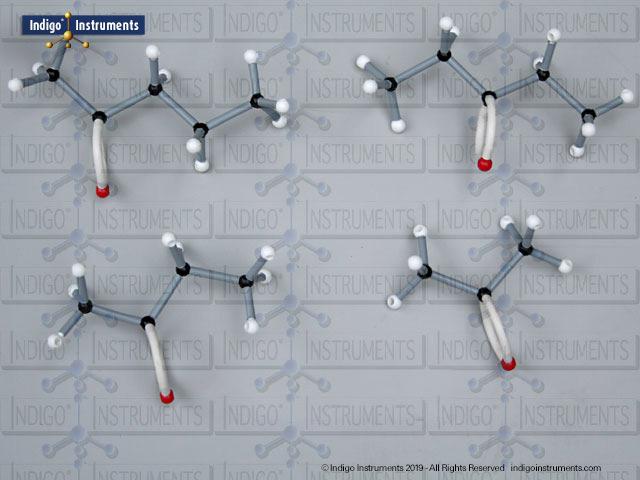
Ether Functional Group Molecule Structure
SKU: 68845NV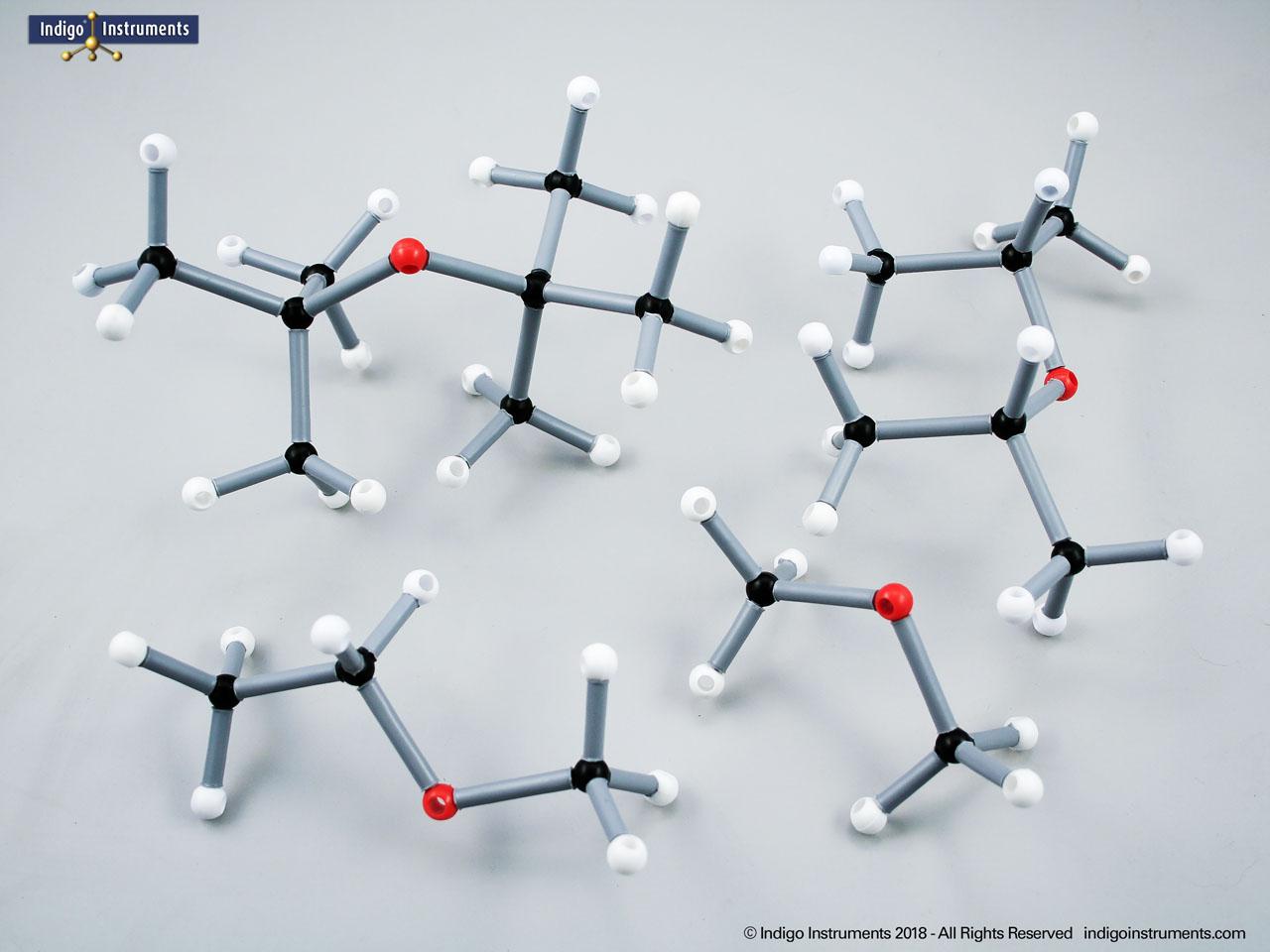
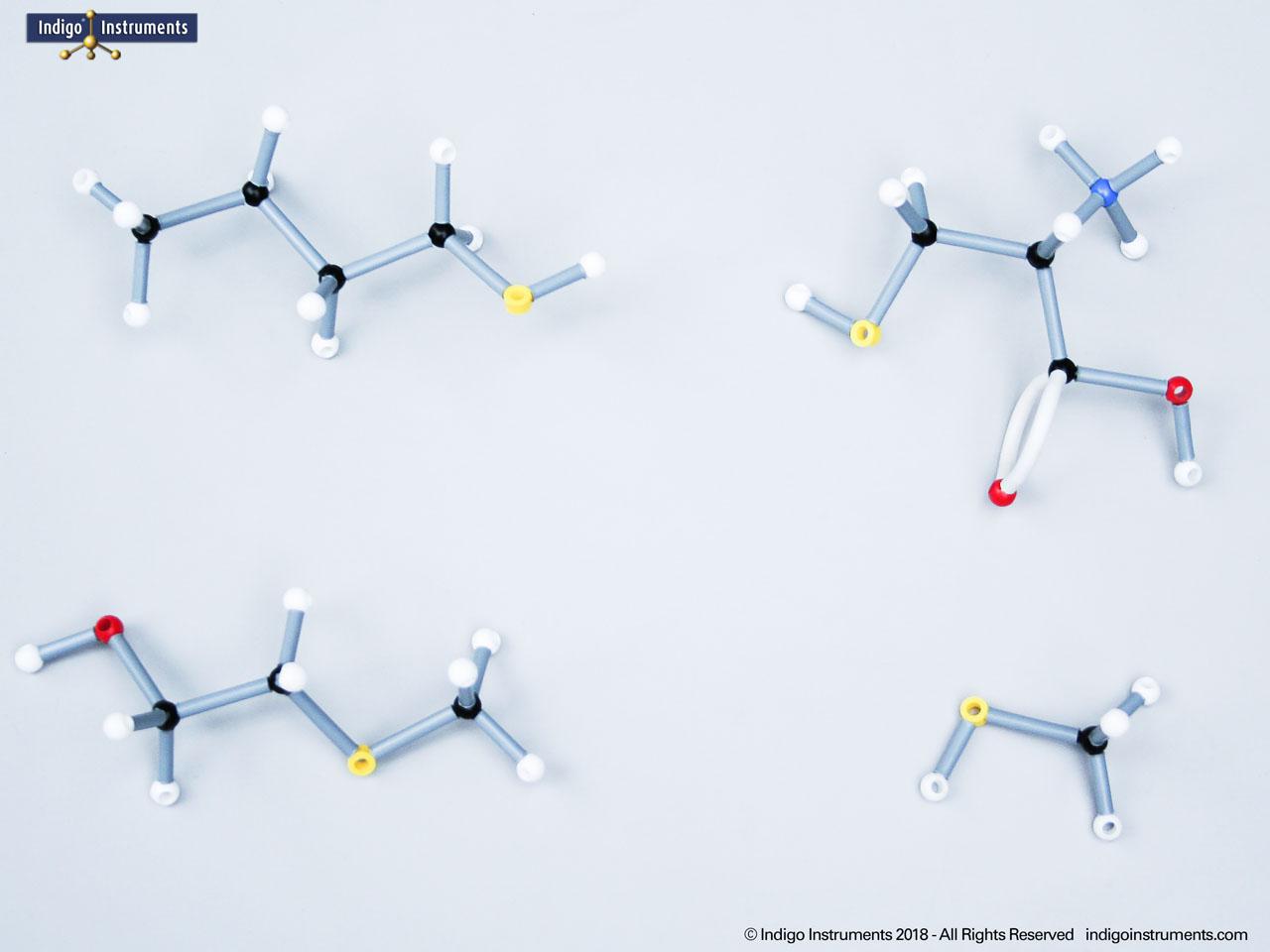
Explore the structure and bonding of ethers with molecular models. Ideal for teaching organic chemistry and biology students how ether linkages influence reactivity, solubility, & biological roles.
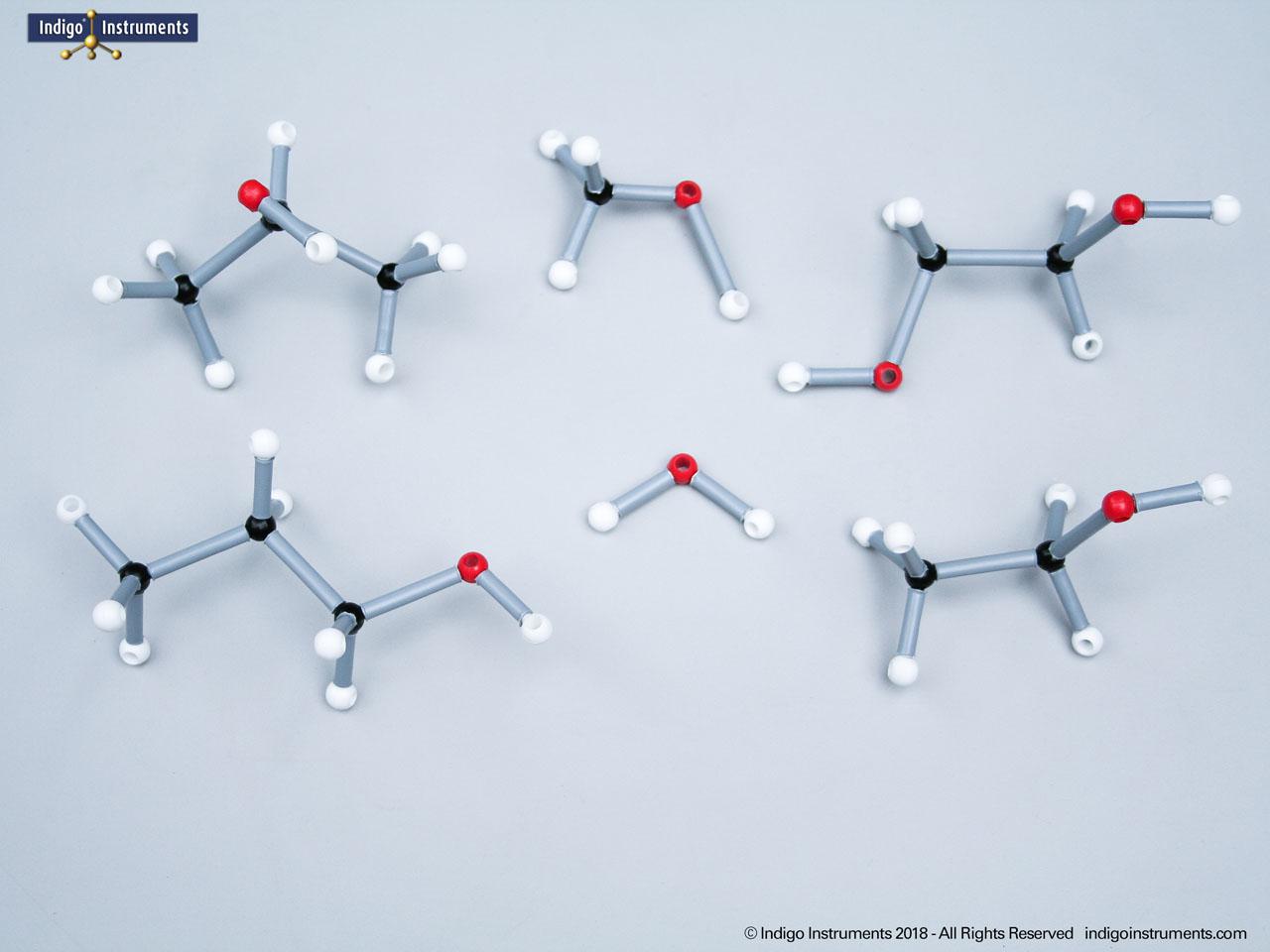
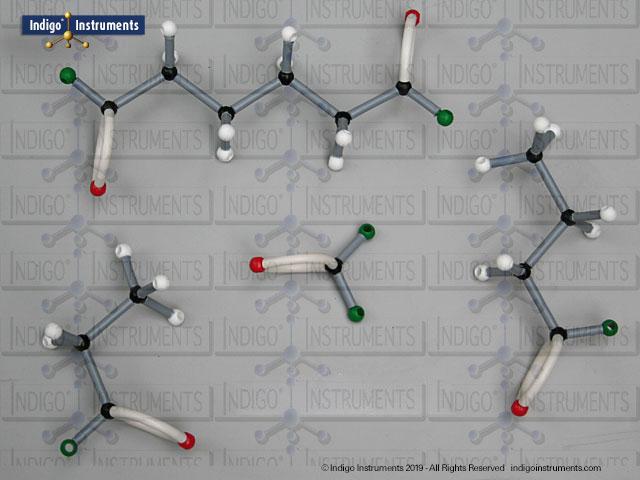
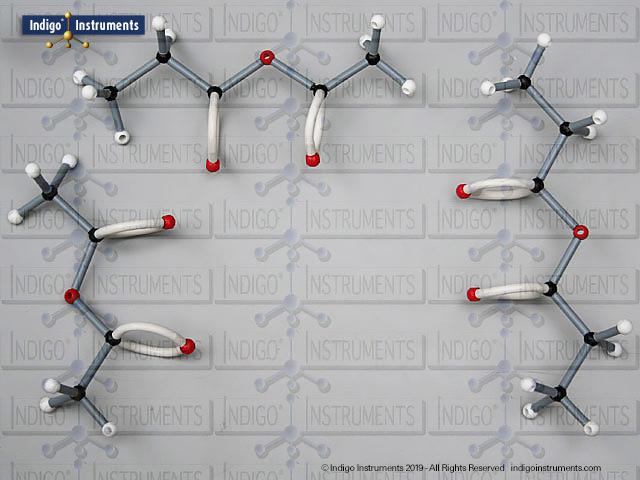
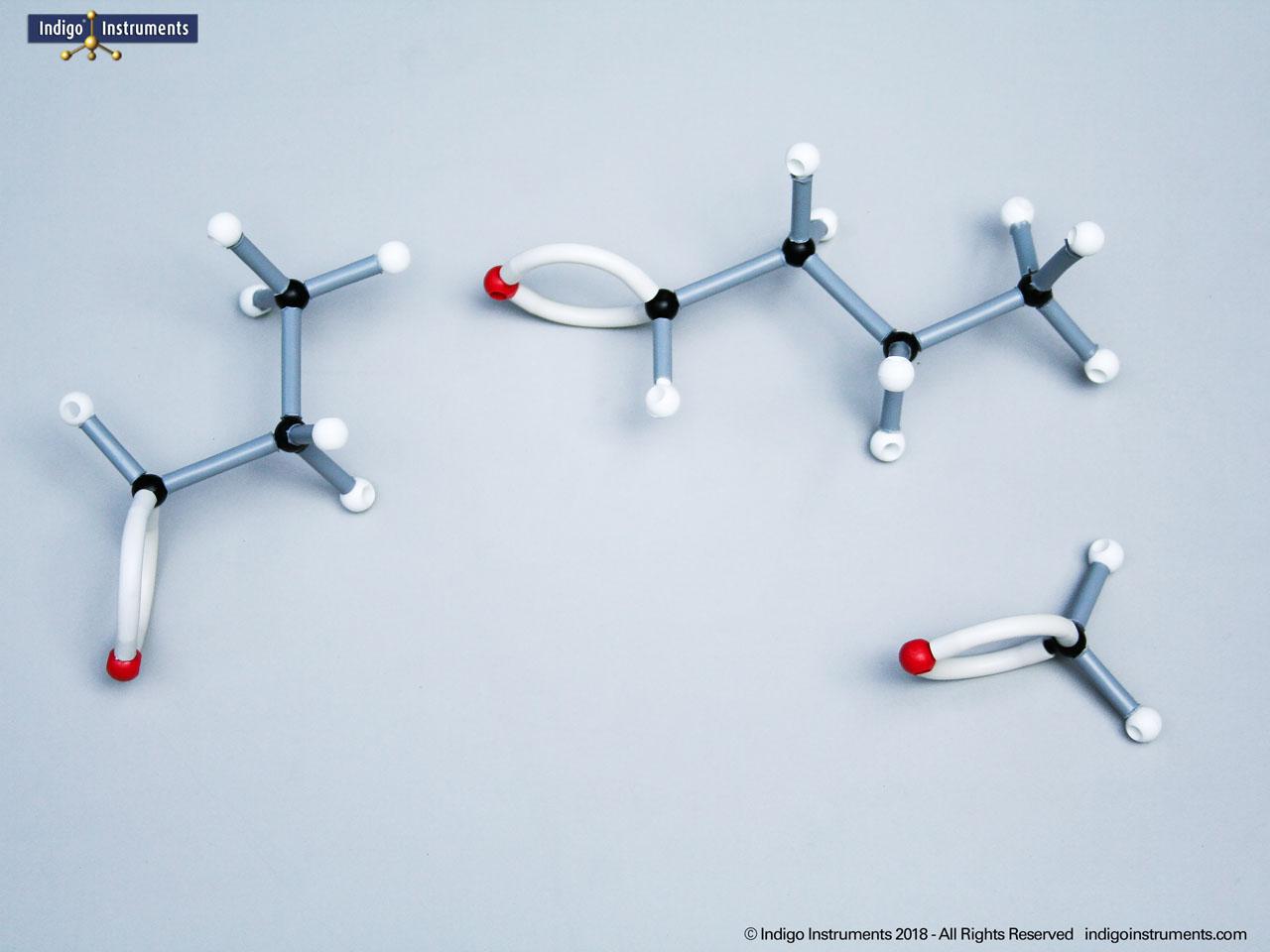
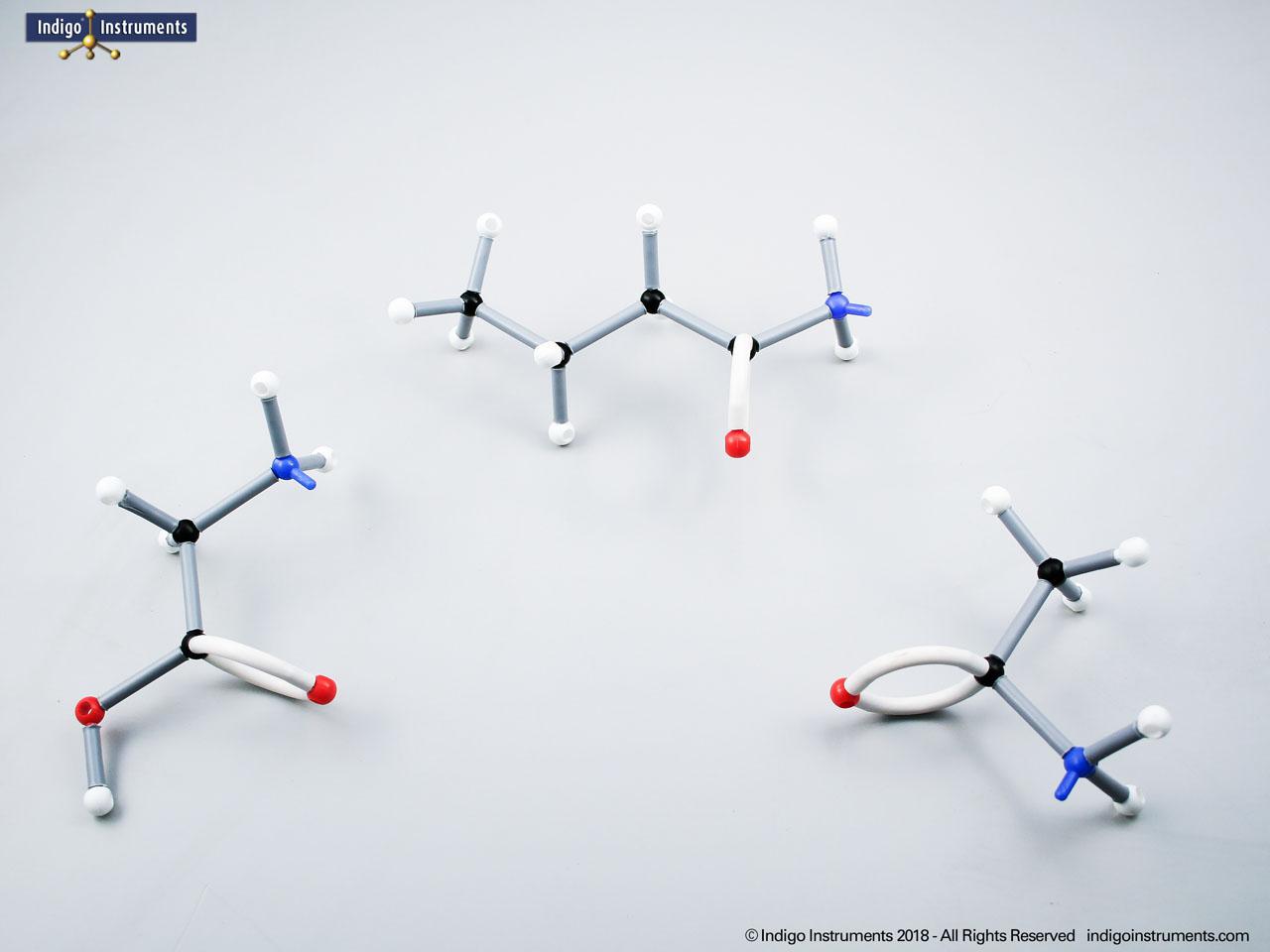
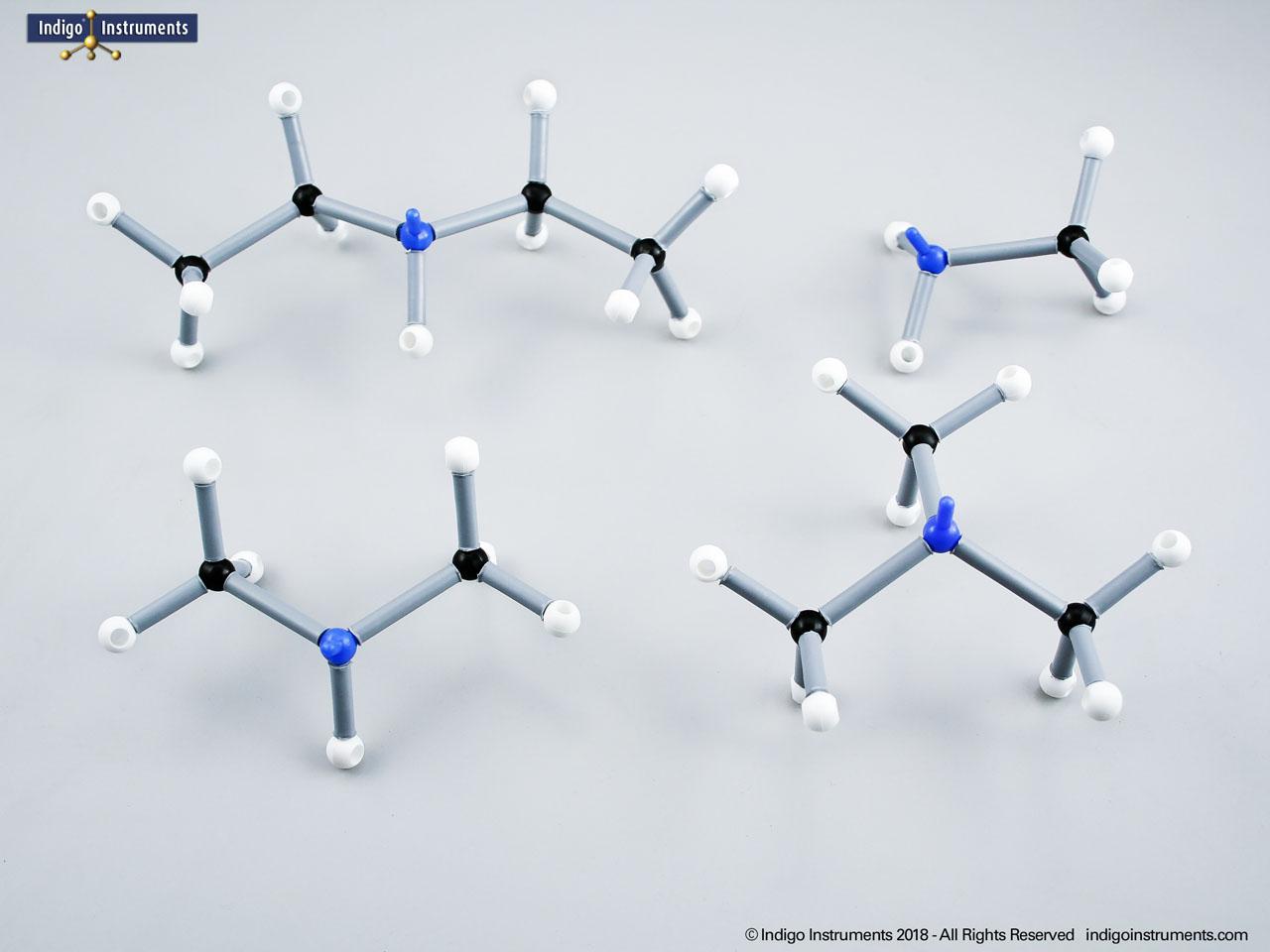
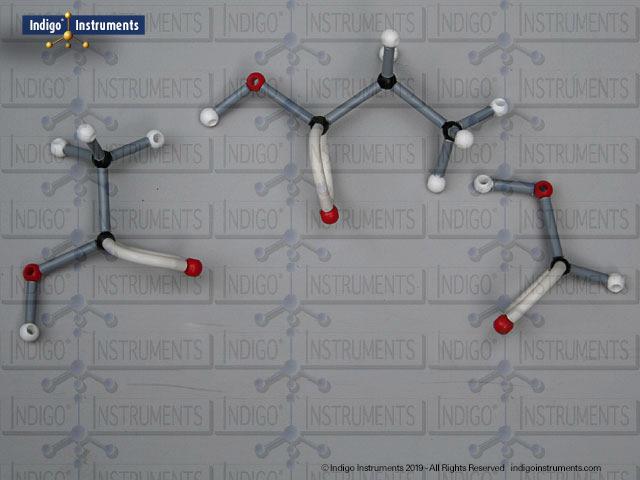
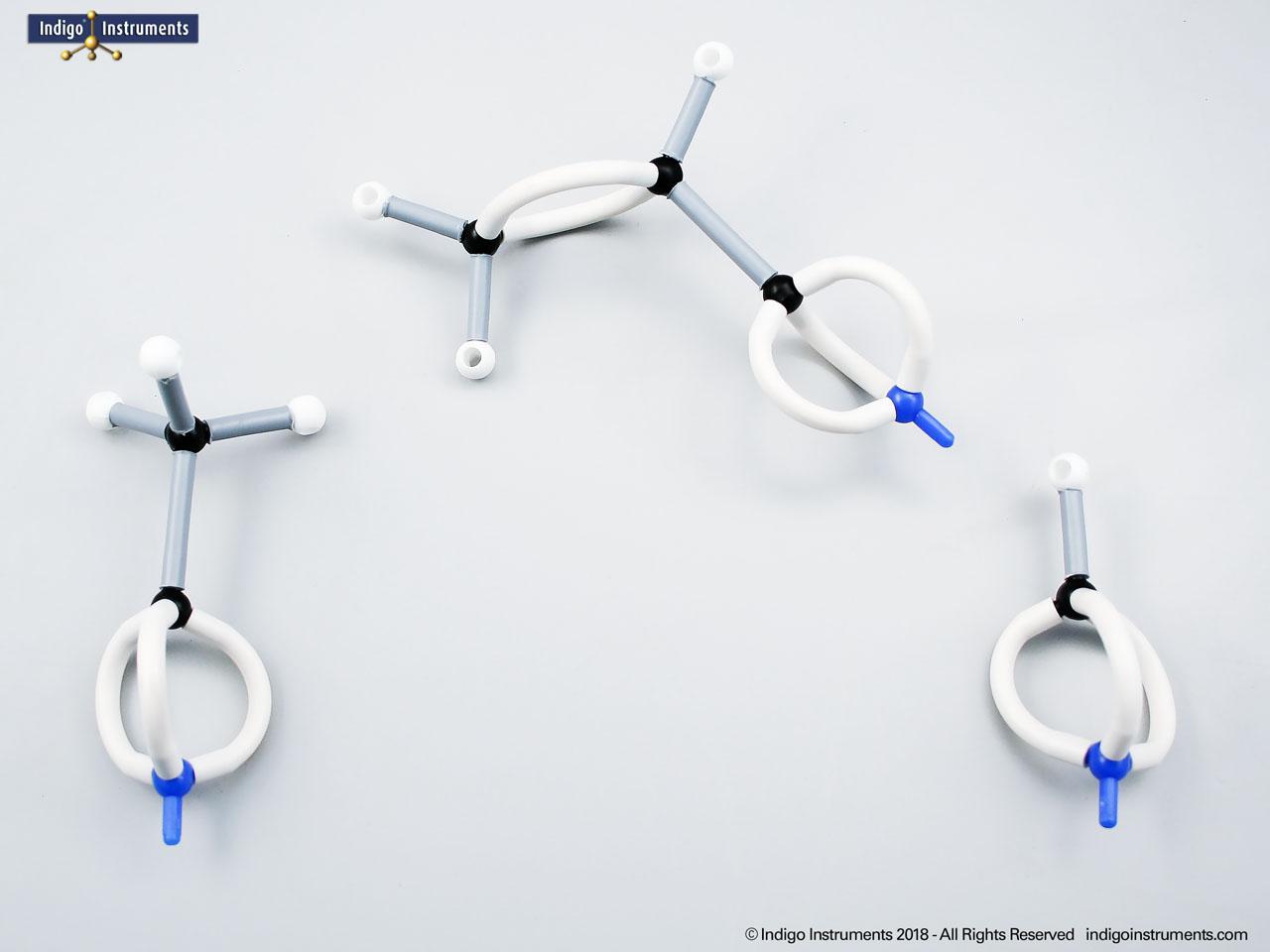
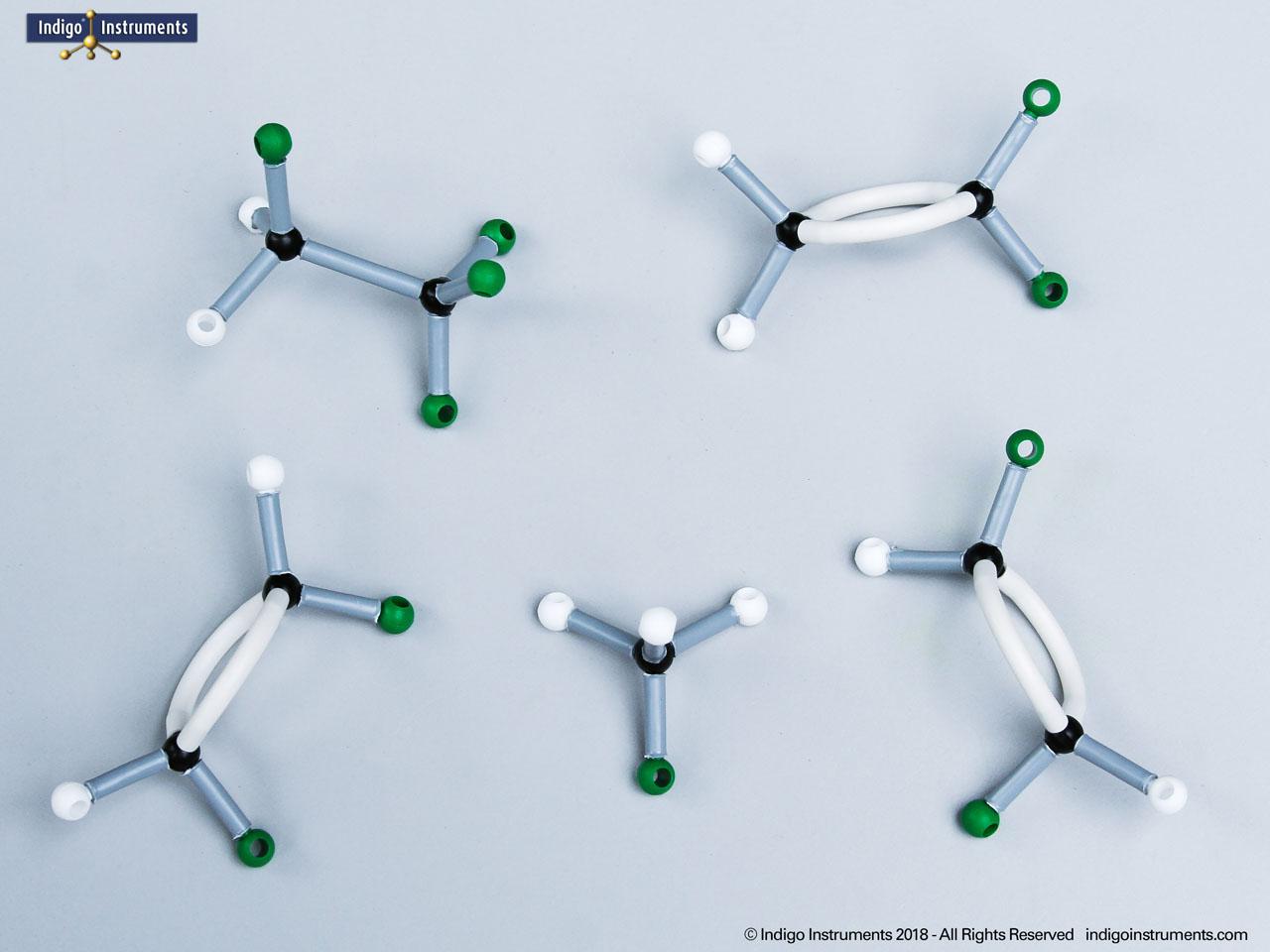
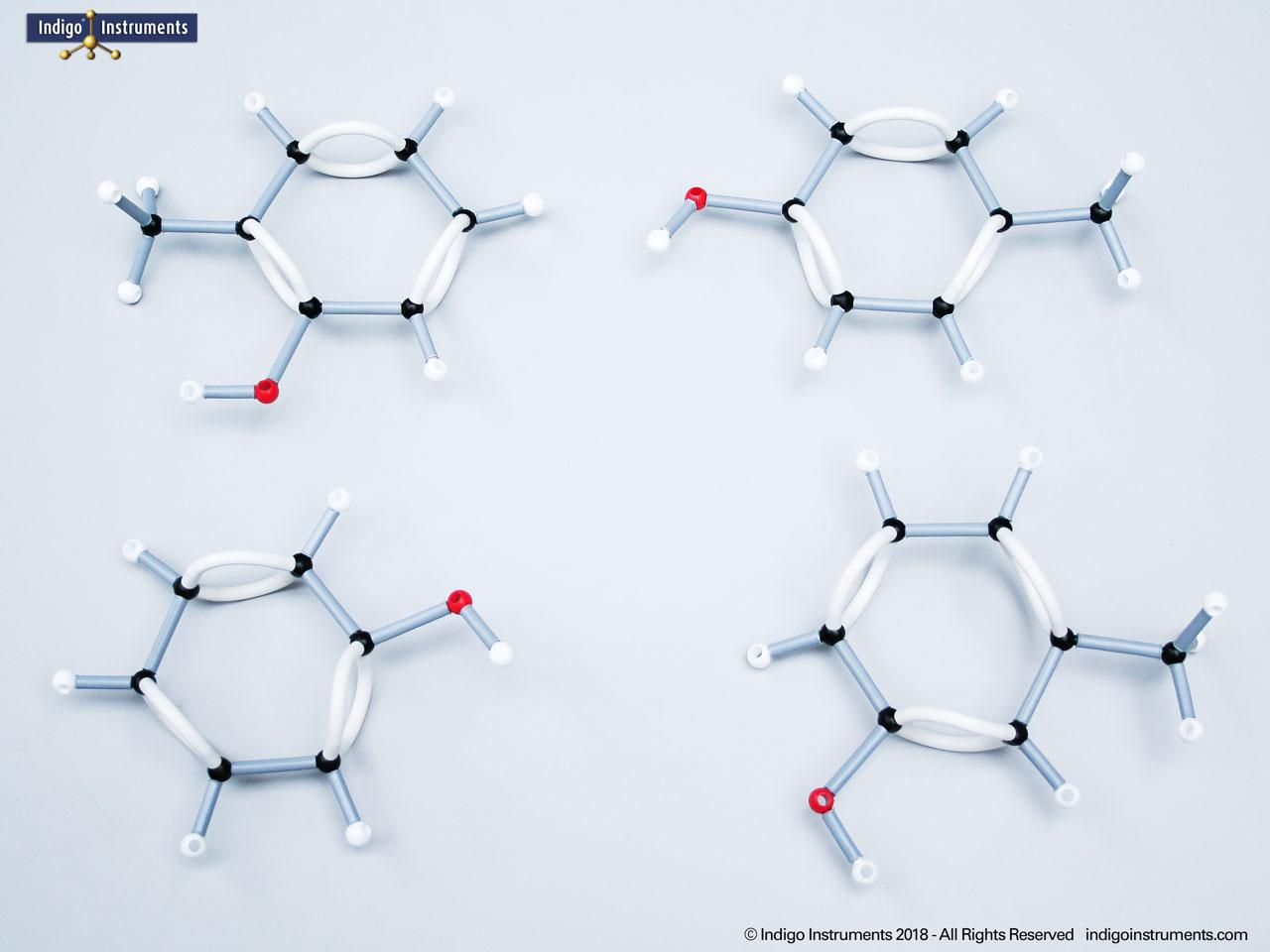
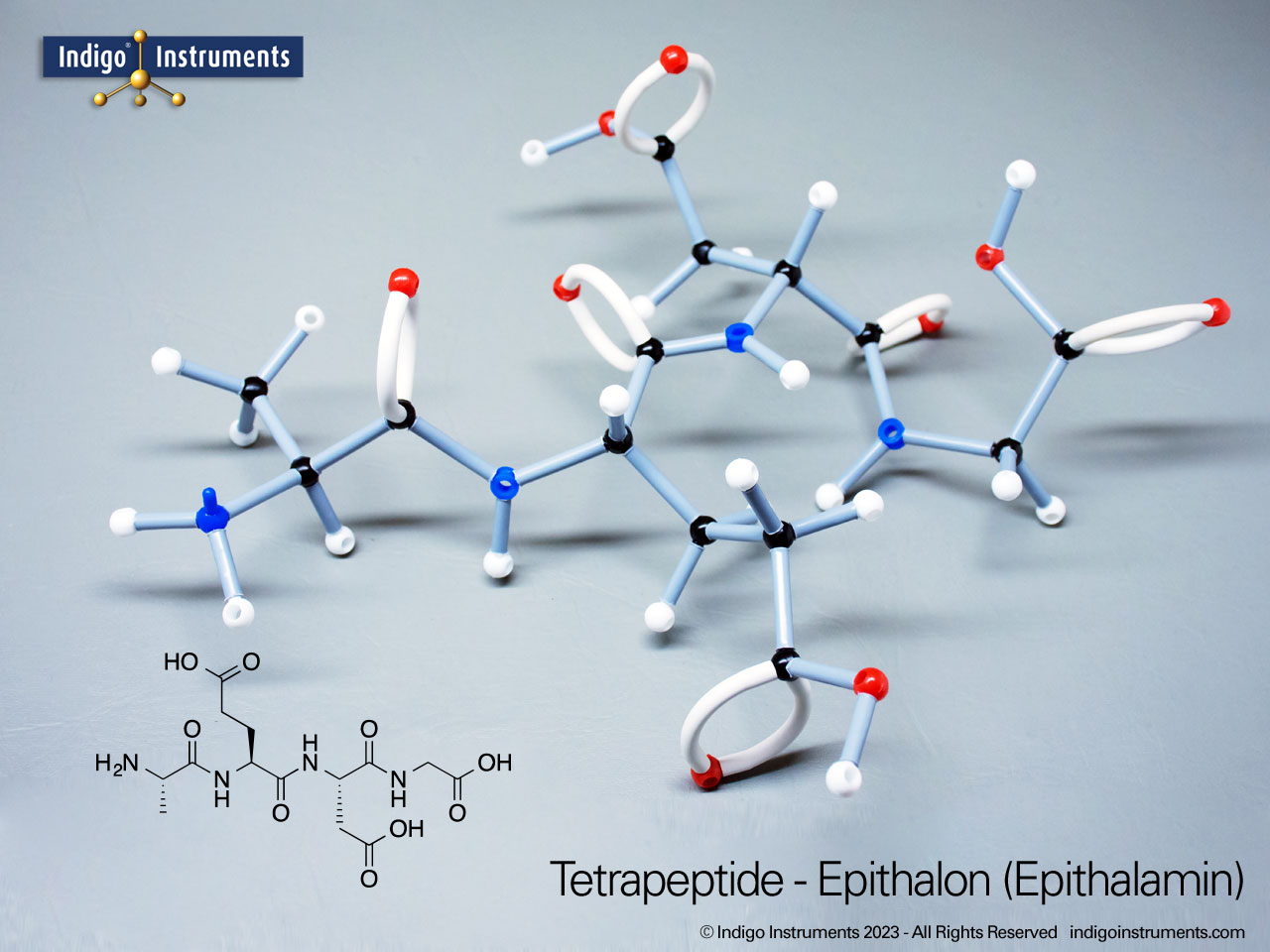
Ethers organic compounds with an oxygen atom bonded to two alkyl or aryl groups (R–O–R′). This oxygen bridge profoundly influences the compound’s polarity, hydrogen bonding capacity, and overall reactivity. In chemistry, ethers are studied for their relatively low reactivity compared to alcohols, their solubility patterns, and their role as solvents. In biology and medicine, ether linkages are found in naturally occurring compounds such as phospholipids and certain anesthetics. These molecular models allow students to visualize bond angles, electron density distribution, and steric effects, and make essential teaching tools for understanding this important functional group.
Indigo Instruments has stocked genuine Cochranes of Oxford molecular model parts for 30+ years. Customize or replenish any model set/kit we have ever sold!