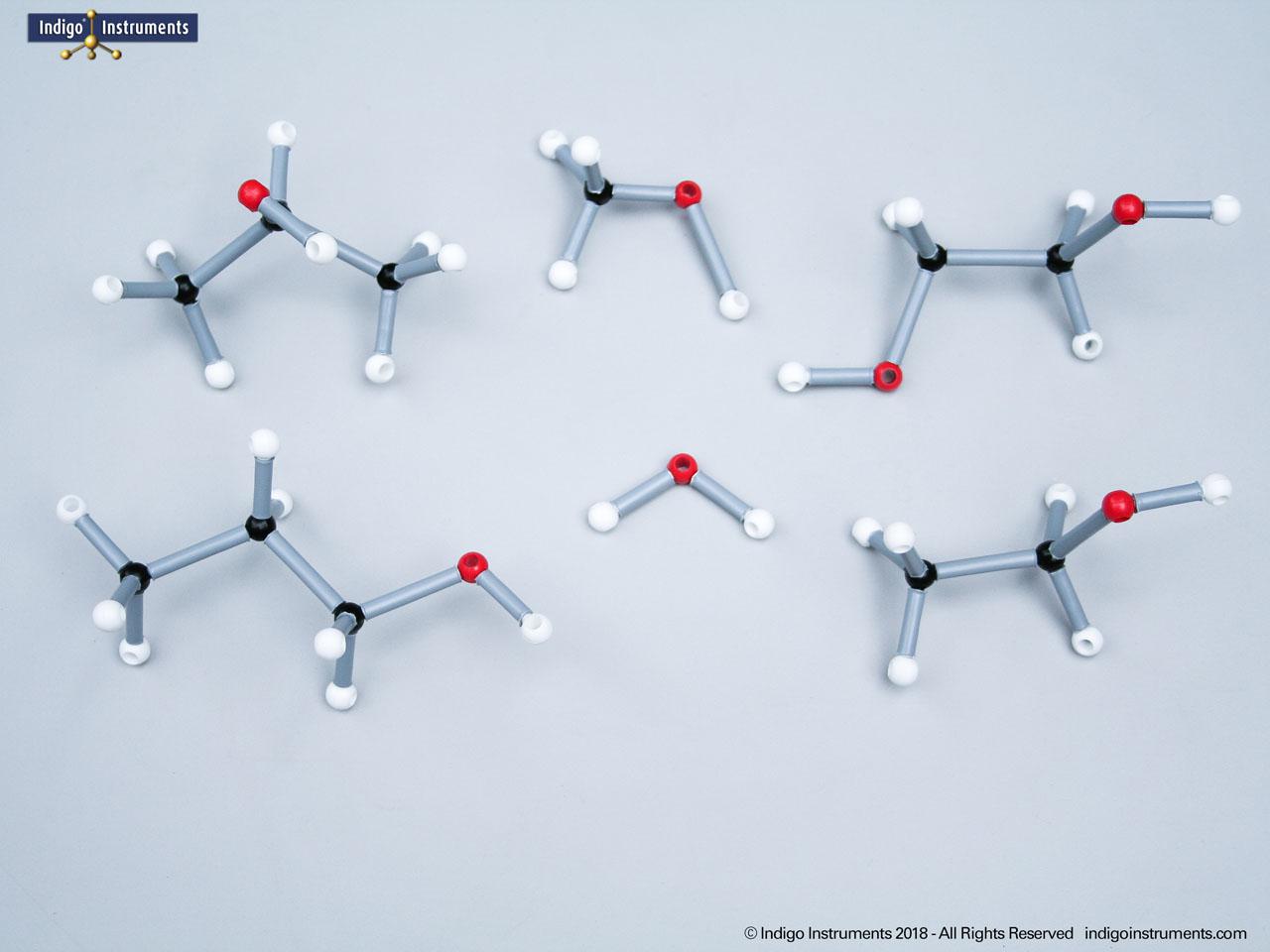
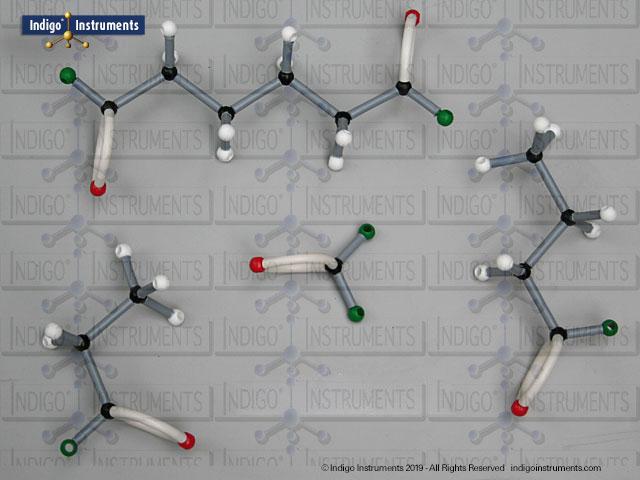
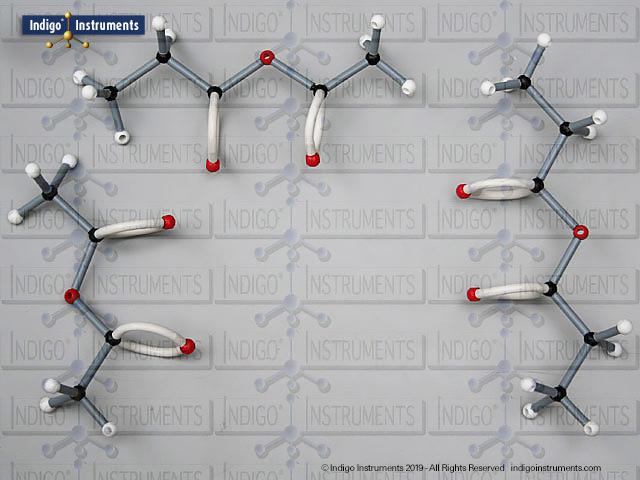
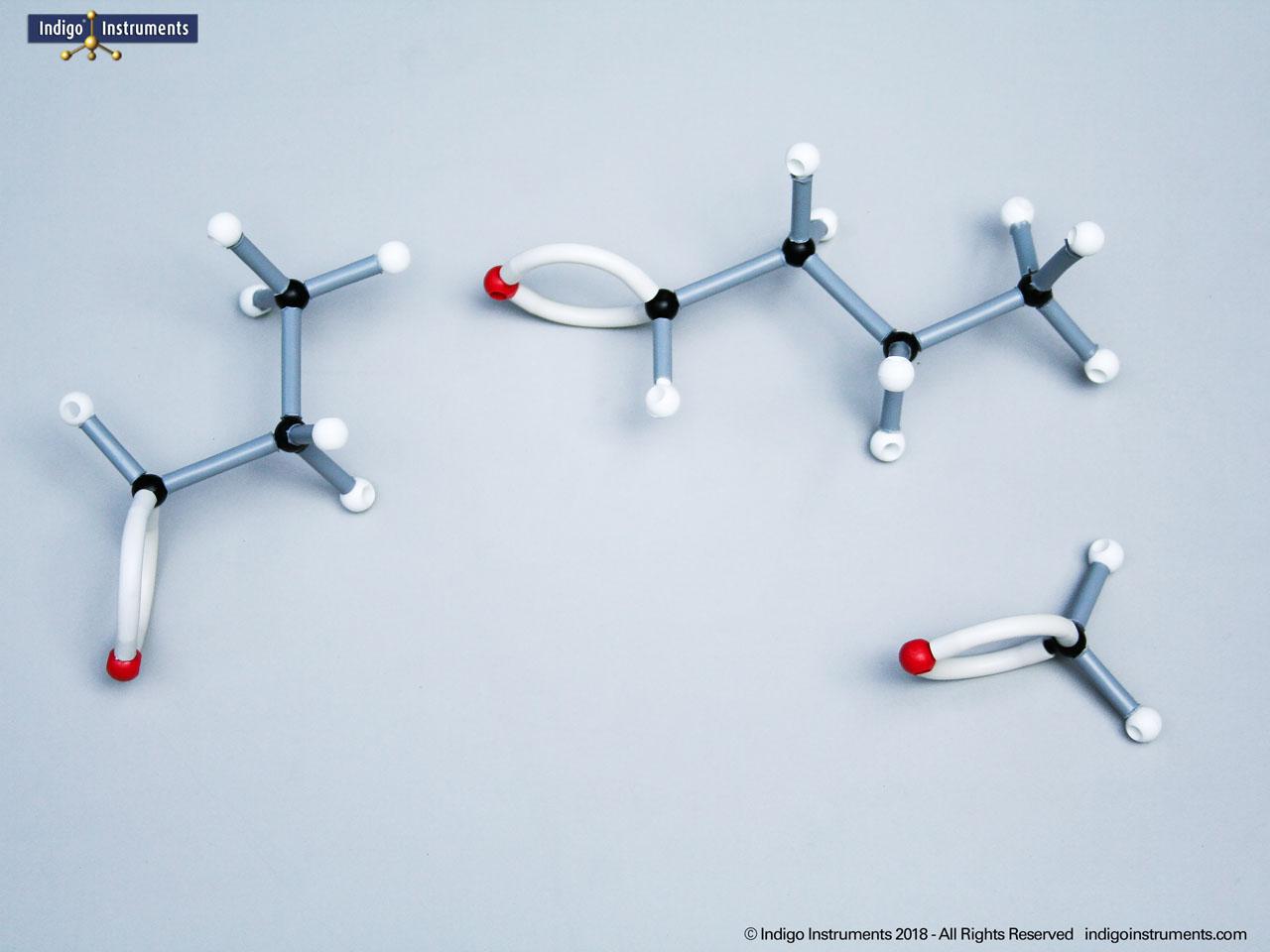
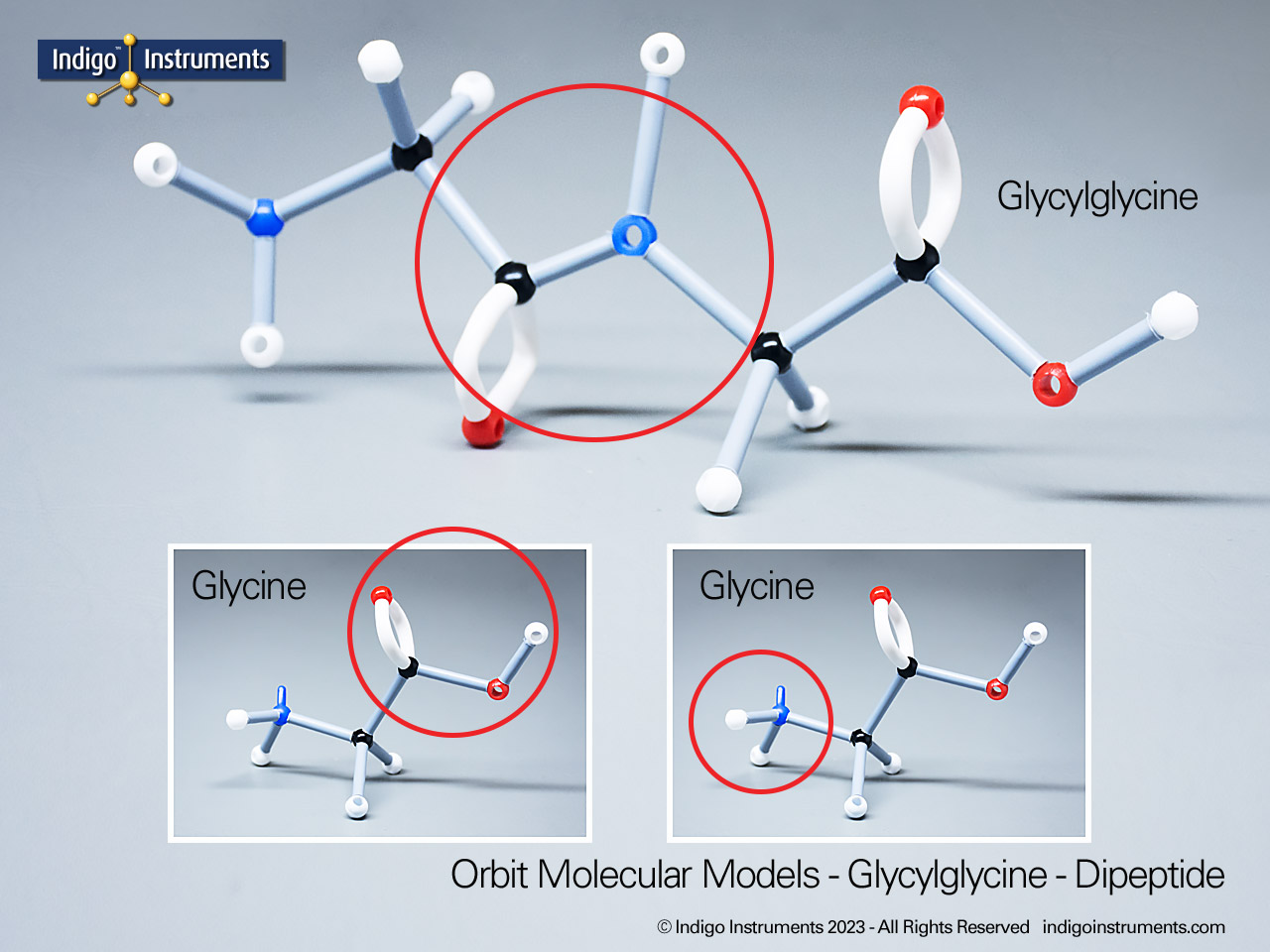
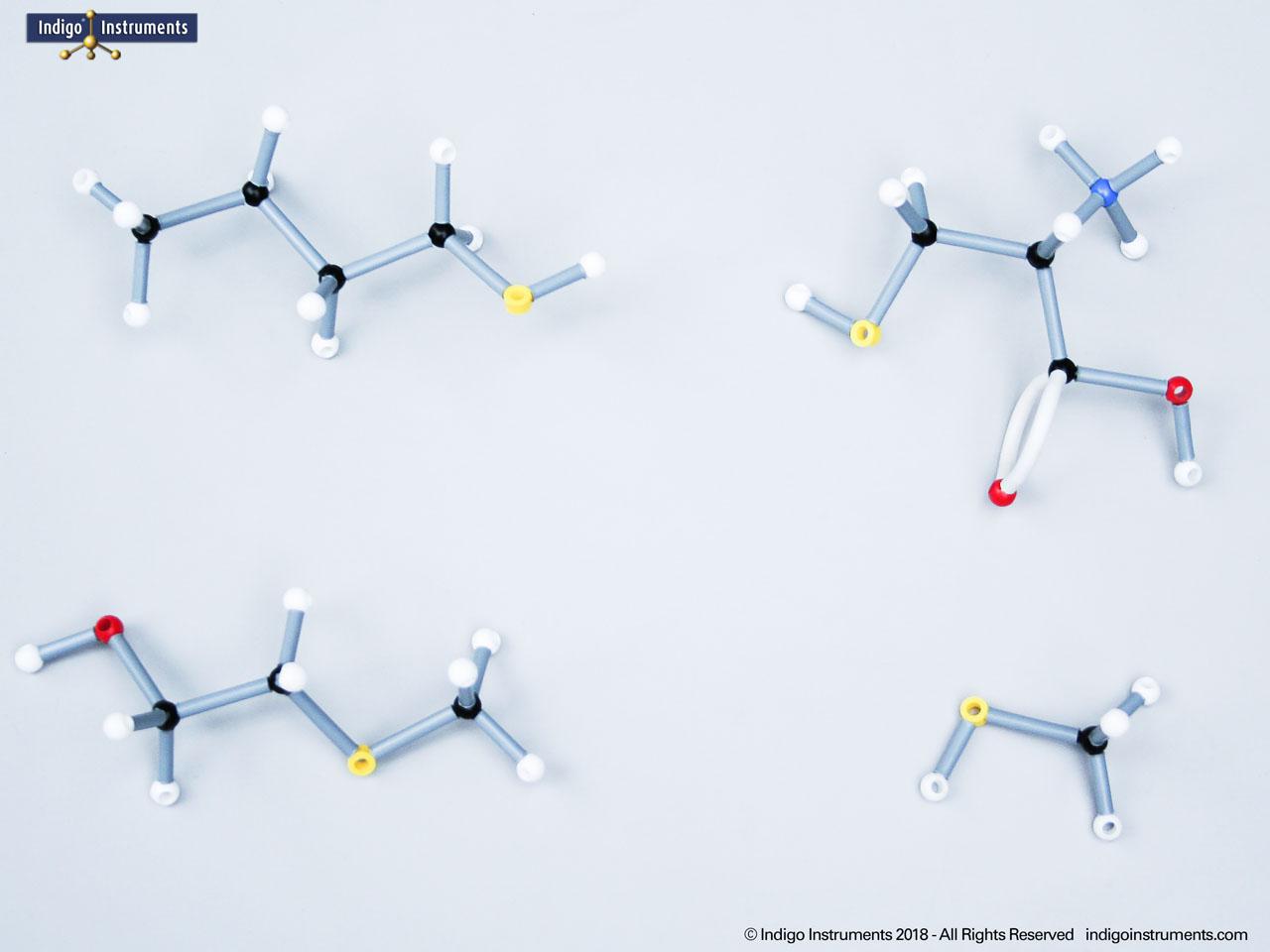
Nitrile Functional Group Molecule Structure
SKU: 68845NV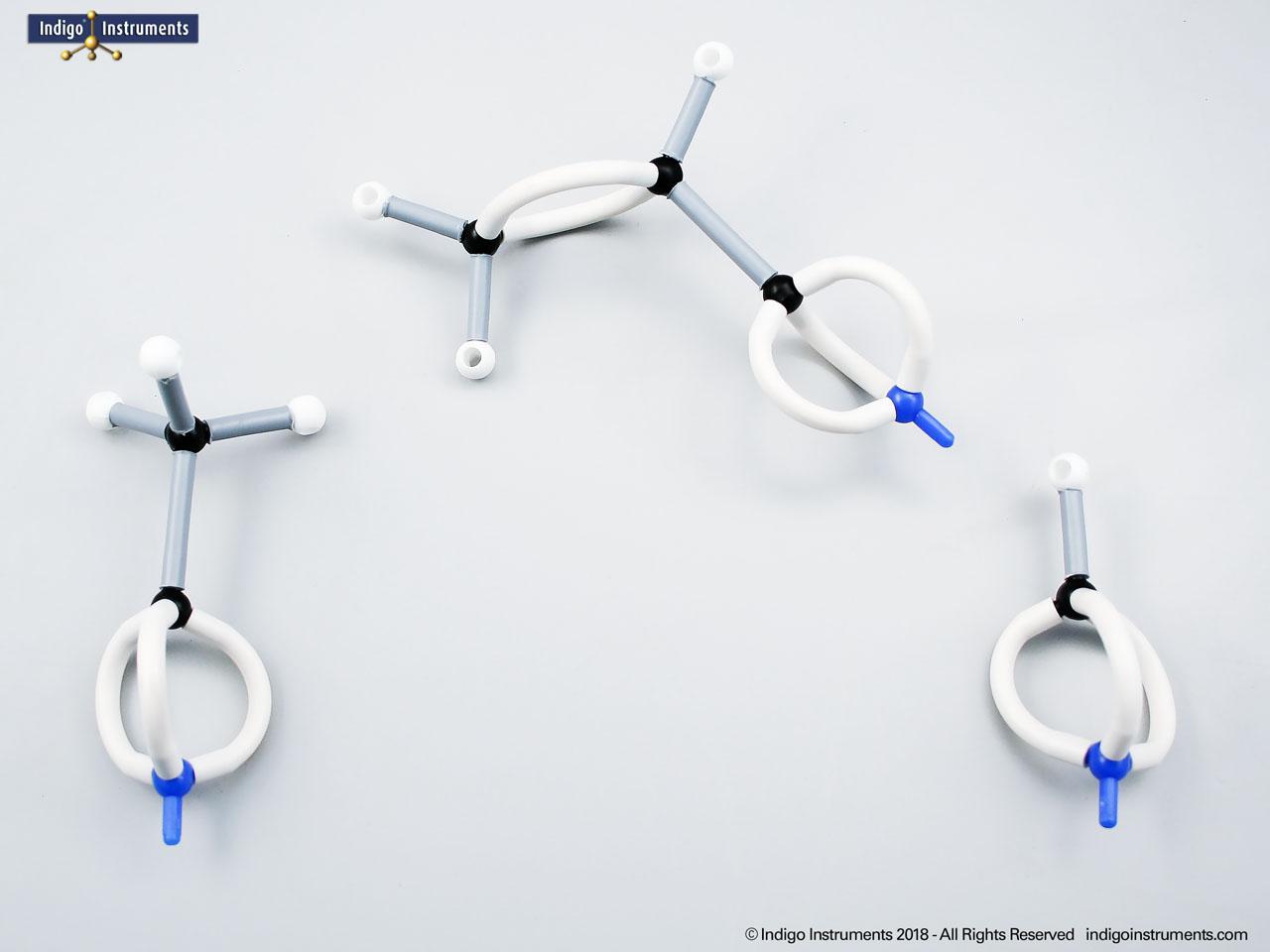
Investigate the nitrile functional group with Orbit molecular models. Learn about key compounds, chemical reactivity, biological roles, and more as they relate to chemical & biological applications.
Nitriles are organic compounds containing the carbon–nitrogen triple bond (–C≡N), a functional group important in both synthetic organic chemistry and biochemical contexts. In chemistry, nitriles are studied for nucleophilic addition, hydrolysis to carboxylic acids, and their role as versatile intermediates in the preparation of amides, amines, and heterocycles. In biology and medicine, nitrile-containing molecules occur in pharmaceuticals, natural products, and metabolic intermediates. Orbit nitrile molecular models allow students to visualize linear geometry, triple-bond electron density, and functional group interactions, making them valuable tools for chemistry majors and biology, nursing, or premed students.
Indigo Instruments has held inventory of genuine Cochranes of Oxford (Orbit) atoms & bonds for 30+ years. These parts are compatible with every molecular model set we have sold since day 1. This quality may appear expensive but no parts support from other vendors costs even more.