Alkane Chemical Compound Models
SKU: 68827W
Build any alkane chemical structure molecular model with the 68827W Advanced Organic Chemistry Model Set from Indigo® Instruments.
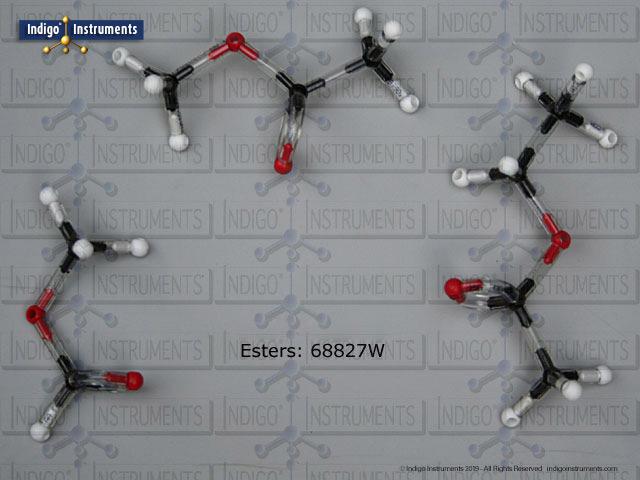
Alkanes are the simplest organic compounds composed of only carbon and hydrogen atoms & are commonly referred to as hydrocarbons. Carbons connect with single covalent bonds in straight chains or as branches & are considered saturated. While not a functional group as such, they are essential to understanding IUPAC namig conventions. The image shows methane, ethane, propane, butane (top right) and isobutane (top left) all of which can be built simultaneously for comparison.
Indigo Instruments has stocked genuine Cochranes of Oxford molecular model parts for 30+ years. Customize or replenish any model set/kit we have ever sold!