Alcohol Functional Group Structure Models
SKU: 68827W
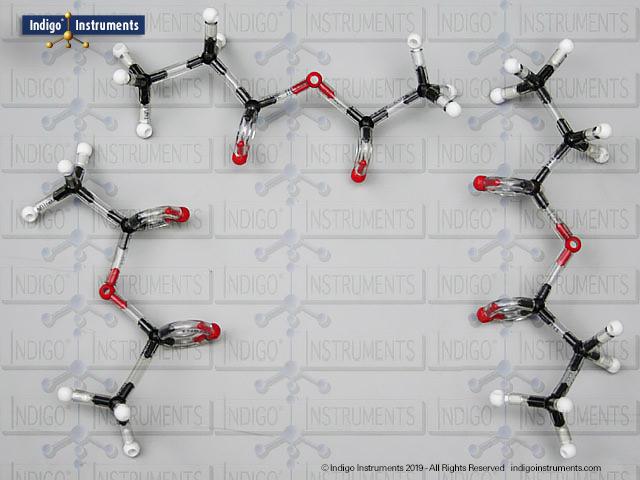
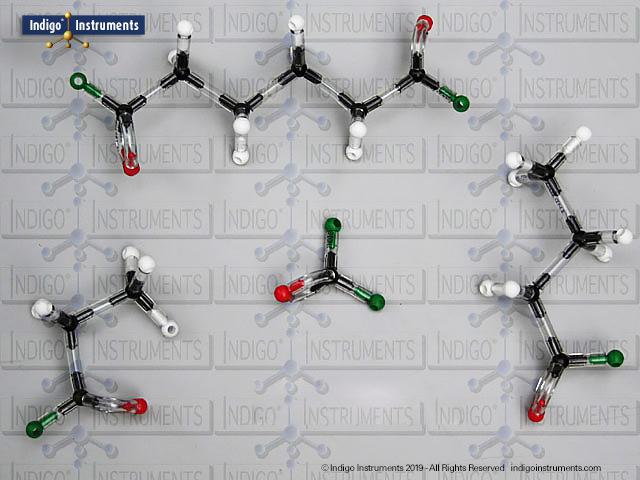
Build any alcohol chemical structure molecular model with the 68827W Advanced Organic Chemistry Molecular Model Set from Indigo® Instruments.
The 68827W Molecular Model Set includes enough carbon, hydrogen & divalent oxygen atoms to build multiple models of alcohols at the same time for side by side manipulation & comparison. Examples include: 2-methyl-1-propanol (primary); 2-methyl-2-pentanol; 2-methyl-2-propanol (tertiary); 2-methylbutan-2-ol; 3, 3, 3-dimethyl-2-butanol (secondary); 5-dimethylhexan-1-ol; 6, 6-methylheptan-3-ol; 8-dimethyl-3-decanol.
Need to build more complex alcohol structures? The Indigo® Instruments 3D Molecular Model Builder can show the structure & list any spare parts you might need to augment this set.
Indigo Instruments has stocked genuine Cochranes of Oxford molecular model parts for 30+ years. Customize or replenish any model set/kit we have ever sold!