NaCl Unit Cell Oh Point Group
SKU: 68791W
Build a sodium chloride unit cell with the Indigo® 68791W molecular model kit to show face-centered cubic packing, Ideal for solid state chemistry, crystallography, materials science, and X-ray diffraction courses.
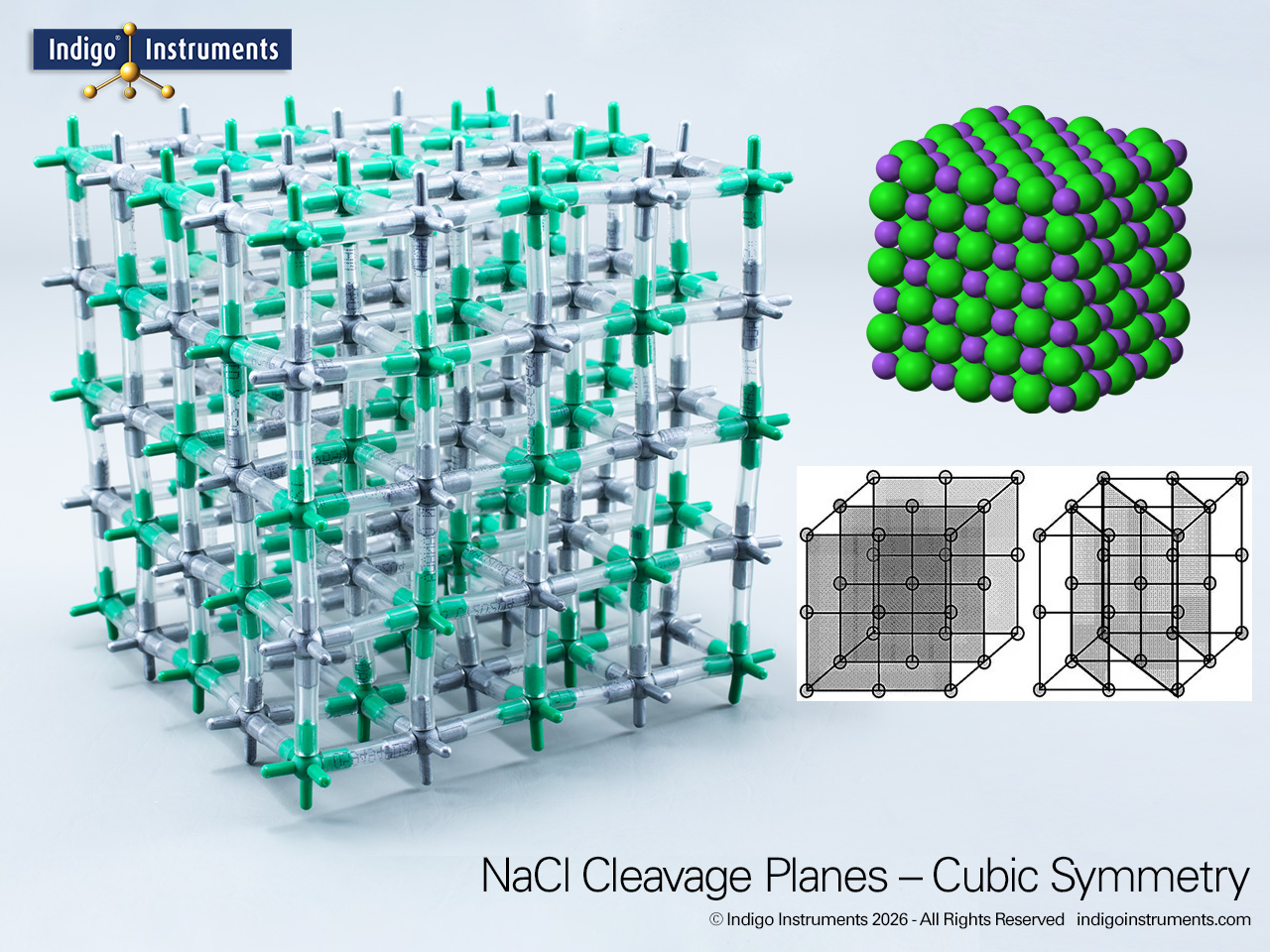
The Indigo® 68791W NaCl model kit can build a sodium chloride (NaCl) unit cell belongs to the Oh point group and crystallographic space group Fm3?m. It shows cubic symmetry of the face-centered cubic lattice where each sodium ion (Na+) is octahedrally coordinated by six chloride ions (Cl-). This 6:6 coordination geometry can also be seen in other rock salt compounds including galena (PbS), periclase (MgO), and iron(II) oxide (FeO).
Indigo Instruments has held inventory of genuine Cochranes of Oxford (Orbit) parts for 30+ years (See Skeletal (Orbit/Minit)) that are compatible with every molecular model we have sold since day 1. This level of quality may appear expensive but no parts support from other vendors costs even more.