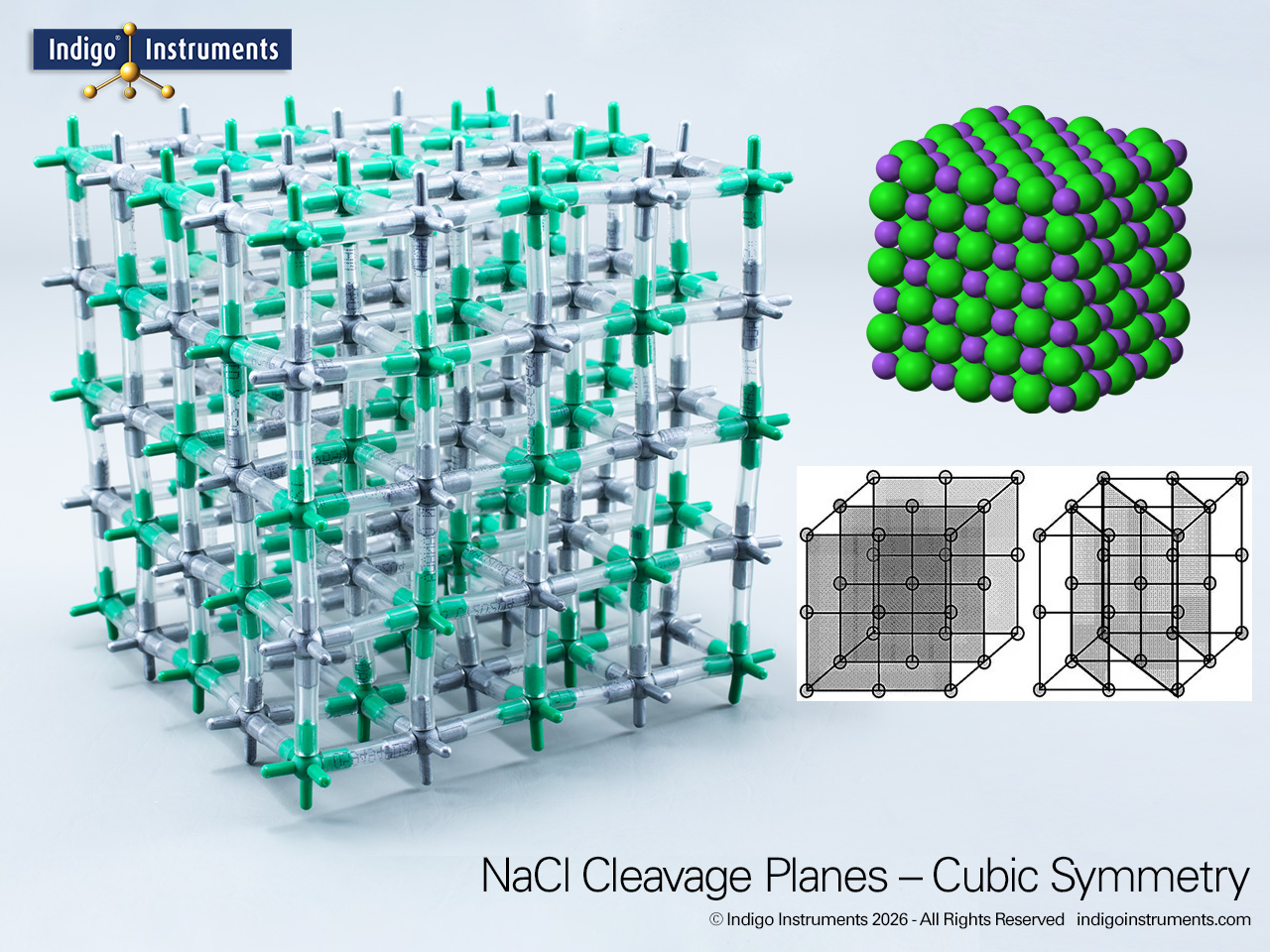
NaCl Crystal Lattice Schottky Defects Model
SKU: 68791W
Learn how point defects in NaCl crystals affect lattice structure, ionic mobility, and conductivity. Explore Schottky and Frenkel vacancies with this hands-on Indigo® NaCl model.
Understanding NaCl point defects is fundamental for solid-state chemistry, materials science, and ionic conduction. Schottky and Frenkel defects explain why ionic solids conduct electricity when molten or in aqueous solution, and why lattice density and mechanical properties vary. Modelling defects helps students visualize vacancy formation, charge balance, and defect migration, and provides a bridge to more advanced topics like semiconductor doping, non-stoichiometric compounds, and defect-driven properties in ceramics.
Indigo Instruments has held inventory of genuine Cochranes of Oxford (Orbit) parts for 30+ years (See Skeletal (Orbit/Minit)) that are compatible with every molecular model we have sold since day 1. This level of quality may appear expensive but no parts support from other vendors costs even more.