NaCl Ionic Lattice Structure Model
SKU: 68791W
Use the Indigo® NaCl ionic lattice model to visualize cubic crystal structure, coordination number, lattice defects and see how atomic-scale geometry defines macroscopic crystal properties. Ideal for chemistry and materials science education.
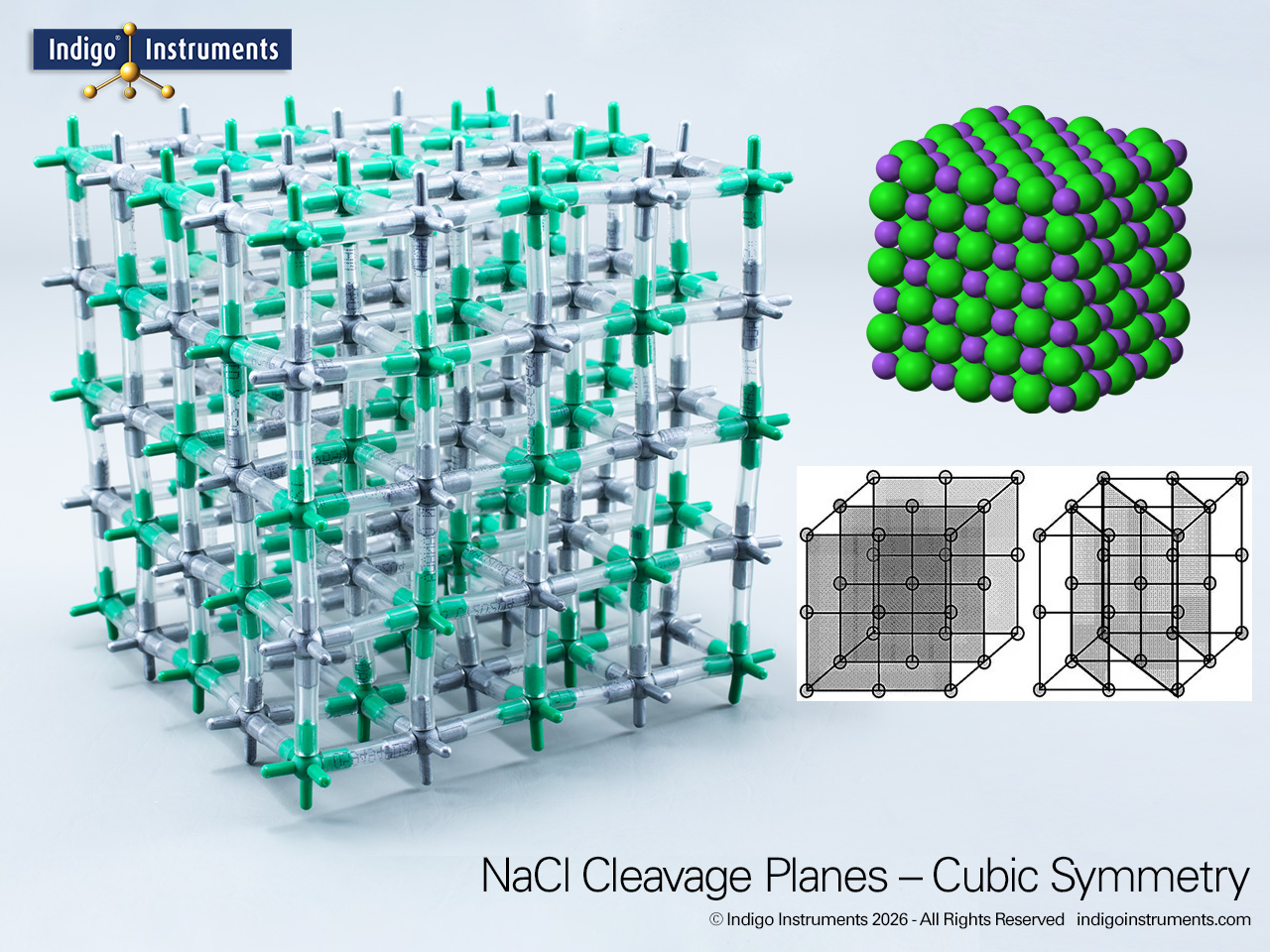
The sodium chloride (NaCl) ionic lattice is an example of a face-centered cubic (FCC) crystal formed by alternating Na+ and Cl- ions. Each ion is surrounded by six of opposite charge producing octahedral coordination found in many simple salts. The resulting cubic symmetry explains the distinctive right-angle cleavage seen in table salt crystals and demonstrates how electrostatic forces and geometric packing govern lattice stability. Building or manipulating this model makes the concept of ionic bonding and coordination geometry tangible for students in chemistry, mineralogy, and materials science.
Indigo Instruments has held inventory of genuine Cochranes of Oxford (Orbit) parts for 30+ years (See Skeletal (Orbit/Minit)) that are compatible with every molecular model we have sold since day 1. This level of quality may appear expensive but no parts support from other vendors costs even more.