NaCl Clevage Planes Model
SKU: 68791W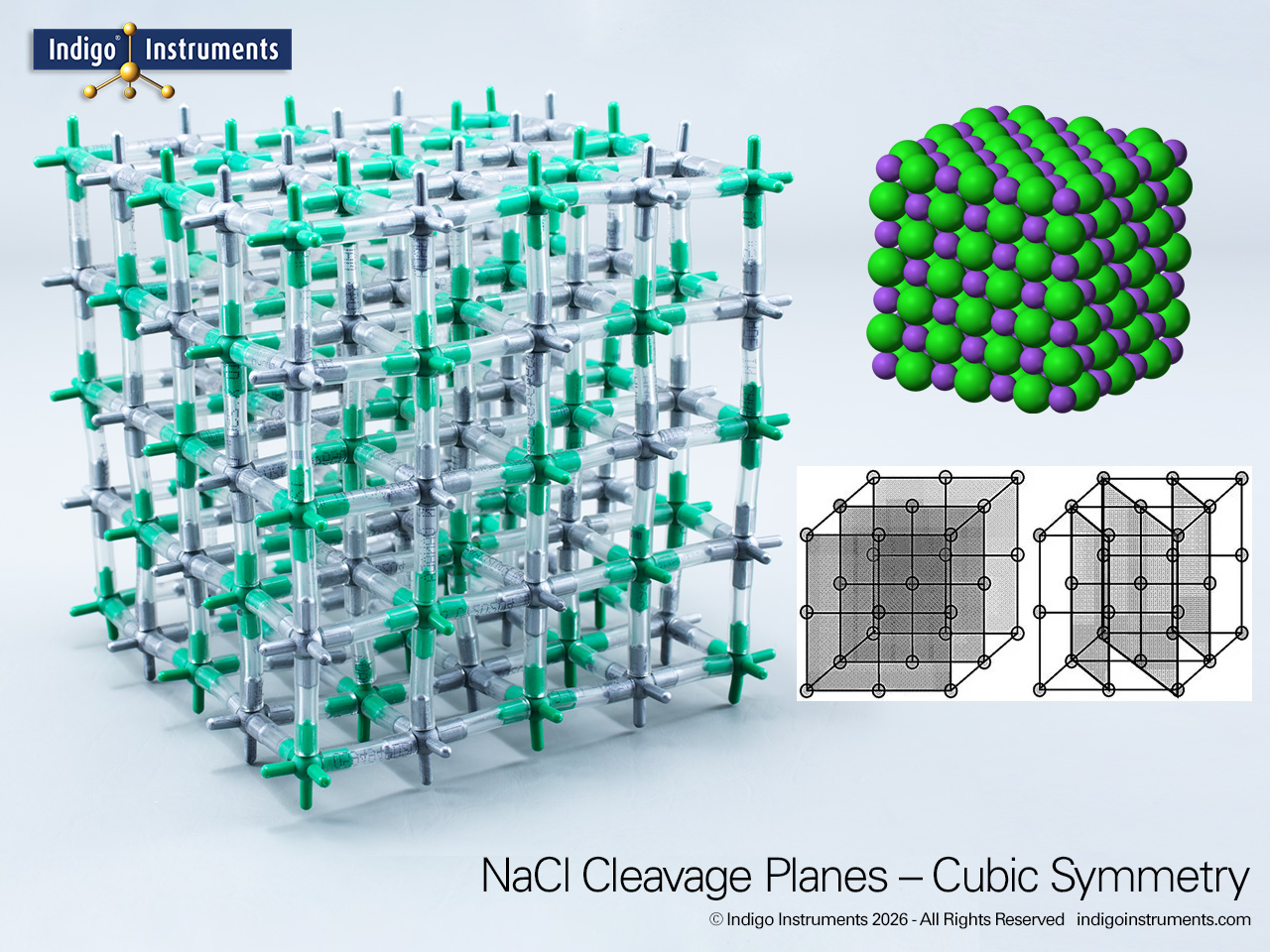
Examine NaCl cleavage planes and learn how ionic arrangement and lattice symmetry determine crystal fracture with an Indigo® NaCl molecular model; ideal for crystallography and chemistry education.
The cleavage planes of sodium chloride (NaCl) reveal the underlying symmetry of its ionic lattice. When struck or stressed, NaCl crystals break cleanly along (100) planes, where each cleavage maintains charge balance by separating layers of Na+ and Cl- ions. These planes correspond to the cube faces of the crystal and reflect the highly ordered face-centered cubic (FCC) arrangement of alternating ions. Demonstrating cleavage with a 5x5x5 atom Indigo® sodium chloride molecular model makes it easy to visualize how electrostatic forces and geometry govern the physical properties of ionic solids.
Indigo Instruments has held inventory of genuine Cochranes of Oxford (Orbit) parts for 30+ years (See Skeletal (Orbit/Minit)) that are compatible with every molecular model we have sold since day 1. This level of quality may appear expensive but no parts support from other vendors costs even more.








