VSEPR Trigonal Planar Geometry
SKU: 69180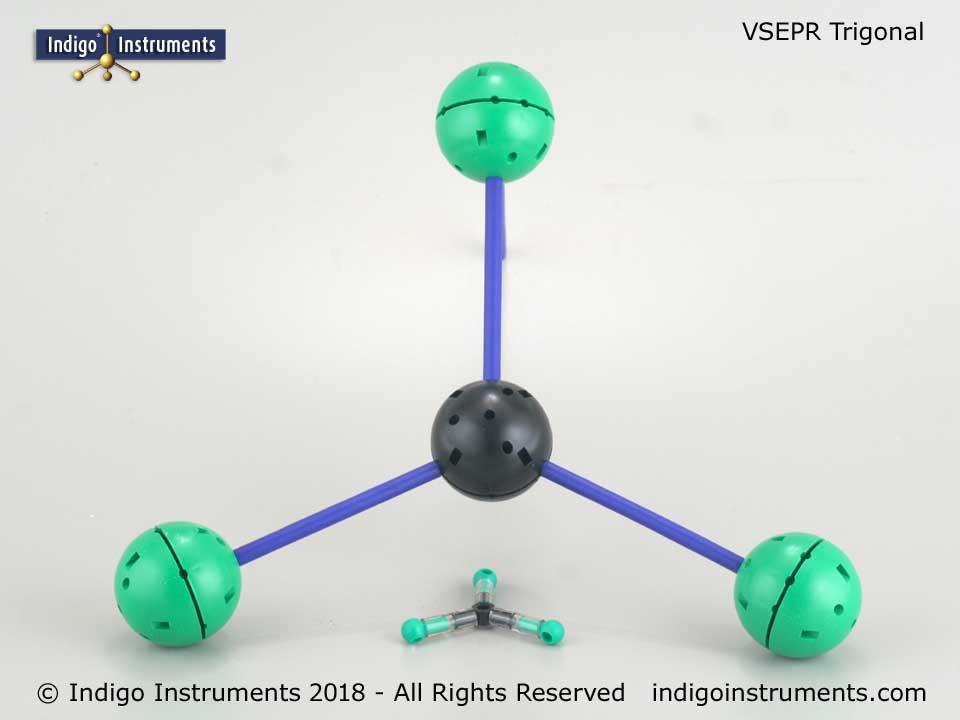
BF3 (boron trifluoride) is a molecule representative of trigonal planar geometry & is roughly 300mm (12") along any 2 arms of the molecule when constructed with Unit models.
The atoms in this VSEPR Large Classroom Model set can assume any geometry. Go to the bottom of page Unit Molecular Models for an overview or click on the Instructions/Safety tab to see videos on how to construct any molecular geometry.
Compare this to the model from the smaller Orbit Basic VSEPR theory set; the same structure is only 50mm (2") along the 2 arms of the molecule.




Thanks for the feedback. It can indeed be a bit tedious setting up the various hole configurations but that is also the beauty of it. No other set is as versatile in creating both standard VSEPR geometries and ones only found in crystals.